|
INTRODUCTION
Gastric cancer remains the second leading cause of cancer death worldwide. Prognosis has improved moderately over the past years but still with very low overall survival rate. The only potentially curative approach for patients with gastric cancer remains early detection and radical surgical resection with curative intent. The overall 5 years survival rate varies from almost nil for patients with metastatic disease to almost fifty percent survival for those patients with early resectable gastric adenocarcinoma.1-6 Patients with localized gastric adenocarcinoma, the 5 years survival is roughly only 10%.1-6 In general, long term survival is closely related to the stage of the disease and its anatomical location.7,8
CASE REPORT
A 50-year-old male was diagnosed with primary gastric lymphoma stage I in 1994. He was then treated with surgical resection in the form of subtotal gastrectomy and billroth anastomosis. In May 2004, follow-up endoscopy showed partial gastrectomy with bile acid induced gastritis and multiple pre-anastomotic polyps probably malignant. Biopsy of gastric and anastomotic area showed findings consistent with chronic active gastritis and well differentiated adenocarcinoma grade I. (Fig. 1)
The endoscopy and biopsy were repeated in March 2005 and November 2005. The histopathlogical findings were consistent with well differentiated adenocarcinoma grade I of the stomach. There was no Helicobacter pylori seen. The malignant cells were positive for cytokeratin (CK). (Fig. 2)
During the period of follow up, patient showed no symptoms and his body weight was maintained at around 62 kg. Computerized axial tomography (CAT) scan of the chest and abdomen revealed no signs of metastatic disease. Tumor markers including CEA, CA125, CA199 and CA153 were normal. CBC, RFT and LFT were also normal.
On day 1, May 2004, the options of treatment were discussed with the patient including observation with best supportive care, surgical intervention, palliative chemotherapy or referral for second medical opinion. The patient declined any type of surgery or chemotherapy. The patient chose observation and regular follow-up.
On March 2010 the patient was evaluated in the oncology clinic and he had no symptoms. His appetite was normal with maintained body weight of 62.5 kg.

Figure 1: H&E shows well differentiated adenocarcinoma of the gastric mucosa Grade I
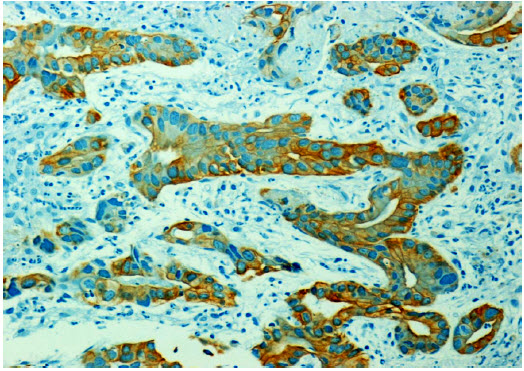
Figure 2: Shows positive immunoreactivity (diffuse and homogenous) of the tumor cells with CK.
DISCUSSION
The histopathologic type of gastric carcinoma include well and poorly differentiated adenocarcinoma, hepatoid adenocarcinoma and denososquamous carcinoma. In the reported patient, the histopathology showed well differentiated adenocarcinoma grade I at the time of presentation and moderately differentiated adenocarcinoma grade II at the time of the last visit 5 years after the first visit.
The only possibility for curative therapy of patients with gastric adenocarcinoma remains surgical resection with free margins. Older patients or patients who had previous gastric surgery have higher postoperative mortality rate. In naïve patient with gastric carcinoma, only 50% undergo gastric resection because of advanced disease and 60% will have either local or distant relapse. In addition, only 30-50% of those who undergo exploration will achieve surgical resection with curative intent.6,9 In one study, the overall survival ranged from one month to 142.5 months, however, less than 5% of patients with gastric adenocarcinoma stage IV will survive 3 years.10 For unresectable advanced or recurrent gastric adenocarcinoma, treatment with systemic chemotherapy improves quality of live (QOL) with marginal survival benefits compared with the best supportive care.11-13 Median survival time of patients with advanced adenocarcinoma of the stomach still remains less than one year.14 Only few patients have survived long period of time longer than five years (2%) with active therapeutic intervention.14
Our patient remains free of symptoms with normal appetite and maintained his body weight for six years. During this period, the gastric adenocarcinoma remained as a local disease with no signs of metastasis; however, the adenocarcinoma has progressed from a grade I to a grade II adenocarcinoma. The reasons why this patient had a local malignant disease without progression over a period of five years could be due to the previous surgical resection effects on the vascular and lymphatic pathway. Secondly, it could be that the malignant disease involved only the superficial mucosa. However, the reason is not clear. At present, one should be aware of such unusual clinical courses of gastric cancer, where prognosis can deviate significantly from the average norm for unknown reasons.
CONCLUSION
This report presents a phenominal case of gastric adenocarcinoma in a patient who is still alive with no signs of metastasis six years after diagnosis without any active therapy.
ACKNOWLEDGEMENTS
The author reported no conflict of interest and no funding was received on this work.
|