Desmoplastic infantile ganglioglioma (DIG) was initially described by Vanderberg in 1987.1 Previously, similar tumors were labeled as composite cerebral neuroblastoma and astrocytoma. This tumor is now recognized as a distinct entity and is included in the World Health Organization classification under the category of pure neuronal and mixed neuronal-glial tumors of the central nervous system.2 Two histological subtypes have been described which include desmoplastic infantile astrocytoma and DIG. DIG has a favorable prognosis and is regarded as a benign grade I tumor.3
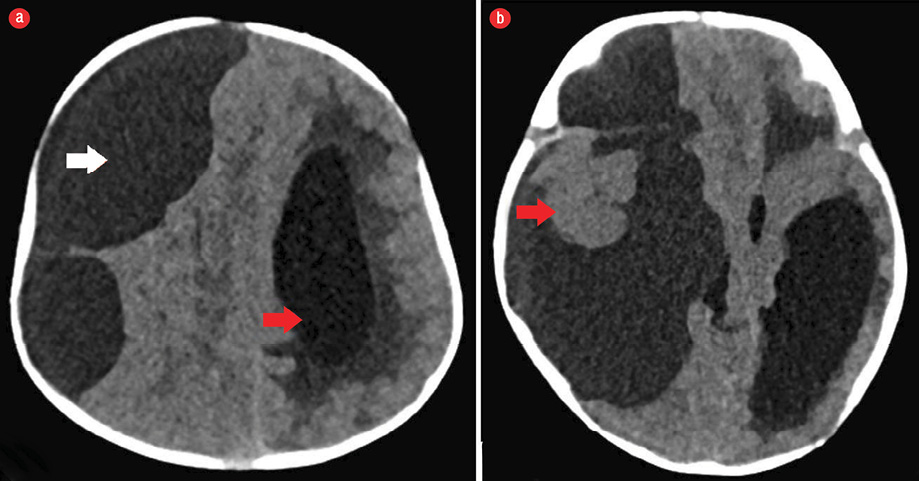
Figure 1: (a) Axial section of non-contrast CT scan of the brain showing a large cystic mass with slight higher attenuation than that of cerebrospinal fluid in the right cerebral convexity (white arrow) displacing the ipsilateral cerebral hemisphere with midline shift and mass effect. Note the dilated contralateral lateral ventricle (red arrow). (b) Axial section of brain non-contrast CT scan at the temporal lobe level showing a subcortical solid component (red arrow), which was overlooked as normal brain tissue at initial presentation.
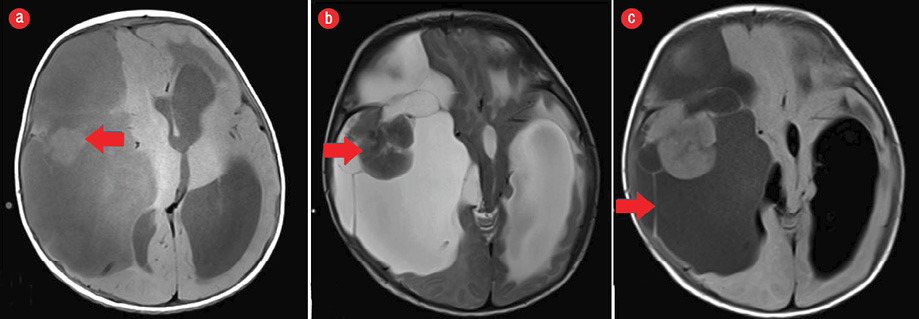
Figure 2: (a) Axial section of non-contrast T1-weighted MRI of the brain revealed a large cystic lesion with higher intensity than that of cerebrospinal fluid. The solid component was isointense to brain parenchyma (red arrow). (b) Axial section of T2-weighted MRI sequence showing the solid mass isointense to the brain parenchyma (red arrow). (c) Axial FLAIR sequence of the brain parenchyma at the same level showing large multiloculated cystic component with multiple thin septations (red arrow).
Case report
A three-month-old infant presented to the pediatric emergency department with excessive crying and decreased activity for three days. No abnormal body movements were noted. The infant was born full-term and delivered via normal vaginal delivery. The family history was unremarkable. Physical examination revealed increased head circumference for the age and an unusual downward gaze preference. Funduscopic evaluation suggested papilledema. A brain computed tomography (CT) scan was ordered to exclude intracranial space-occupying lesions and hydrocephalus.
Plain CT scan of the head [Figure 1] showed a supratentorial large cystic attenuating mass with density little higher than cerebrospinal fluid (CSF). The mass was causing a significant mass effect and compressing the right cerebral hemisphere of the brain parenchyma. It was causing effacement of the right lateral ventricle, midline shift, and secondary dilation of the contralateral left lateral ventricle. Mild displacement and rotation of the brainstem was seen. A small component isodense to the brain parenchyma at the level of the temporal lobe was overlooked because it simulated the normal brain parenchyma with mass effect. The CT scan findings were in favor of an extra-axial cystic mass causing significant mass effect. Thus the differential diagnosis of a huge arachnoid cyst was forwarded considering the CT scan findings.
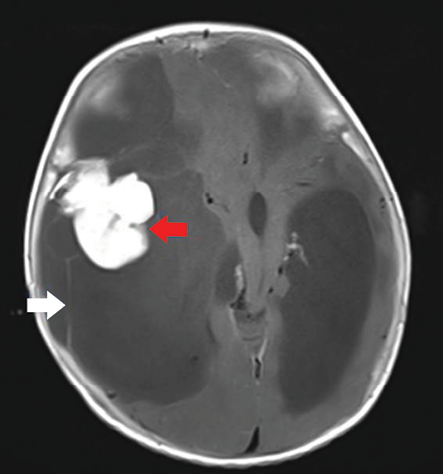
Figure 3: Axial section of post-contrast T1-weighted image show avid contrast enhancement of the solid component (red arrow) and mild enhancement of the septations (white arrow).
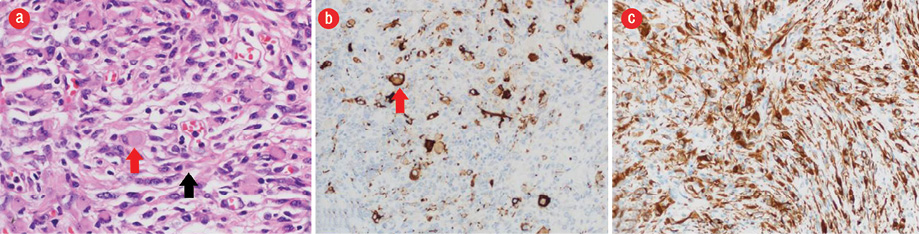
Figure 4: (a) Hematoxylin and eosin stain showing desmoplastic component with fibroblast-like spindle cells (black arrow) intermixed with slightly pleomorphic astrocytic cells with eosinophilic cytoplasm (red arrow) arranged in fascicles demonstrating storiform pattern. (b) Synaptophysin stain highlights few scattered neuronal cells (red arrow) in the glial component. (c) Diffuse and strong positive immunostaining of the glial component and weak staining of the poorly differentiated component for glial fibrillary acidic protein. Magnification = × 100.
Magnetic resonance imaging (MRI) of the brain [Figure 2] was done for specific characterization of the mass. MRI revealed an extensive right-hemispheric multilobar (frontoparietotemporal) space-occupying mass, with a predominant multiloculated cystic component, which was incompletely suppressed on FLAIR sequence. The mass was peripherally abutting the meningeal surface. An eccentric, peripherally located lobulated solid component was abutting the dura showing intermediate intensity on T1- and T2-weighted images, with no diffusion restriction on diffusion-weighted imaging. The lobulated soft tissue component showed intense post-contrast enhancement after the administration of gadolinium [Figure 3]. There was slight enhancement of multiple hairline septations. The solid-cystic mass was causing significant mass effect with contralateral midline shift, subfalcine, and uncal herniation. The MRI features were characteristic of DIG or a cystic low-grade astrocytoma.
The patient underwent complete resection of the tumor. Histopathology sections of postoperative specimens suggested a glioneuronal tumor composed of a dominant desmoplastic leptomeningeal component and poorly differentiated neuroepithelial component [Figure 4]. The desmoplastic component consisted of fibroblast-like spindle cells intermixed with slightly pleomorphic astrocytic cells with eosinophilic cytoplasm arranged in fascicles or demonstrating storiform pattern. Few neuronal cells and gemistocytes were also present. The other component consisted of poorly differentiated neuroepithelial cells with small round deeply basophilic nuclei, minimal cytoplasm, and frequent mitosis.
Immunohistochemical staining showed strong positive immunostaining of the glial component and weak staining of the poorly differentiated component for glial fibrillary acidic protein. Synaptophysin highlights a few scattered neuronal cells in the glial component. Ki-67 was approximately 4% proliferative index in the glial component and approximately 60% in the poorly differentiated neuroepithelial component. Thus, the final diagnosis was DIG. Total resection of the tumor was the treatment of choice and was performed when the patient was three-months-old. The papilledema disappeared postoperatively, and at one-year follow-up, no tumor recurrence was seen, and the patient had no neurological deficits.
Discussion
DIG is a rare supratentorial neuroepithelial tumor that comprises 0.5–1.0% of all intracranial tumors. A meta-analysis documented that only 94 cases of DIG have been reported in the literature so far.4 The tumor is characterized by its large size, desmoplasia with a cystic component, and a solid astrocytic/ganglionic differentiation. DIG usually presents before the age of 18 months, with the median age of diagnosis between five and six months old.5 However, cases have been reported in adolescents and young adults.6 Males are affected more frequently than females. Symptoms are predominantly due to the effect of the mass, which includes seizures, paresis, increased head circumference, or bulging fontanels.7,8
DIGs present as a large mass with cystic and solid components. The cystic component can be unicystic or multicystic, usually located deep inside the lesion and the solid component is located peripherally in the subcortical location often adjacent to the meninges.5 These tumors can grow massively and manifest with relatively few symptoms. DIG invariably arises in the supratentorial region and commonly involves more than one lobe, preferentially the temporal and frontal lobes.5–8
On CT scan, the cystic component is hypodense with attenuation close to that of CSF, and the solid component usually enhanced after iodinated contrast injection. There is no communication between deep cysts and the lateral ventricles.9 Meningeal enhancement and thickening adjacent to the solid portion of the tumor may be seen.9 Features of bone abnormalities adjacent to a tumor consisting of skull bossing, thinning, and deformation are reported in the literature.4–9 On CT scan, DIG appears as large hypodense cystic masses with a solid isoattenuating or slightly hyperdense superficial portion,4 and as a hypointense cystic mass with an isointense peripheral solid component on T1-weighted MRI. The peripheral solid component avidly enhances following gadolinium injection.4,5,9 On T2-weighted images the cystic component is hyperintense, and the solid component usually appears isointense to the cortex. Perilesional edema is usually absent or if at all present will be mild.
The radiological differential diagnosis of DIG includes primitive neuroectodermal tumor, ependymoma, ganglioglioma, and dysembryoplastic neuroepithelial tumor.10 Primitive neuroectodermal tumor also appear as solid-cystic mass at this age and usually have a prominent cystic component, but the tumor lies deep in the white matter and solid component is less peripheral.10 Supratentorial ependymoma has predilection for parietal lobe, but with a paraventricular location.9,10 Ganglioglioma is usually located within the temporal lobe.9 It is smaller than the DIG and contains both a solid and cystic component. Dysembryoplastic neuroepithelial tumor is usually a cortical supratentorial tumor mass without perilesional edema. Mass effect is absent, and a cystic component is occasionally found.11
Histologically, the most prominent feature of DIG is desmoplasia and spindle cells with a storiform pattern of arrangement.7 There is also a ganglion cell component, which is present as single cells or clusters.12 Treatment is essentially surgical with no complimentary treatment needed in cases of complete tumor resection.13
In our case, non-contrast CT showed a large cystic tumor involving frontal, parietal, and temporal lobes. The isodense solid component was overlooked because it was difficult to differentiate it from the normal brain parenchyma. However, the MRI features were characteristic, especially after contrast administration and helped to guide the diagnosis. Pathology of the tumor revealed typical histological features. Our patient underwent complete resection of the tumor without any postoperative complications.
Conclusion
Our case extends the reported spectrum of this rare tumor and will help radiologists in considering the diagnosis of a large solid-cystic mass especially in infants. The MRI features are characteristic for the preoperative diagnosis of DIG which has a favorable prognosis. The small solid component may be overlooked in CT and the mass might be mistaken as an arachnoid cyst; however, MRI features are characteristic for the diagnosis of DIG.
Disclosure
The authors declared no conflicts of interest.
references
- 1. VandenBerg SR, May EE, Rubinstein LJ, Herman MM, Perentes E, Vinores SA, et al. Desmoplastic supratentorial neuroepithelial tumors of infancy with divergent differentiation potential (“desmoplastic infantile gangliogliomas”). Report on 11 cases of a distinctive embryonal tumor with favorable prognosis. J Neurosurg 1987 Jan;66(1):58-71.
- 2. Shin JH, Lee HK, Khang SK, Kim DW, Jeong AK, Ahn KJ, et al. Neuronal tumors of the central nervous system: radiologic findings and pathologic correlation. Radiographics 2002 Sep-Oct;22(5):1177-1189.
- 3. Bhardwaj M, Sharma A, Pal HK. Desmoplastic infantile ganglioglioma with calcification. Neuropathology 2006 Aug;26(4):318-322.
- 4. Gelabert-Gonzalez M, Serramito-García R, Arcos-Algaba A. Desmoplastic infantile and non-infantile ganglioglioma. Review of the literature. Neurosurg Rev 2010 Apr;34(2): 151-158.
- 5. Per H, Kontaş O, Kumandaş S, Kurtsoy A. A report of a desmoplastic non-infantile ganglioglioma in a 6-year-old boy with review of the literature. Neurosurg Rev 2009 Jul;32(3):369-374, discussion 374.
- 6. Ganesan K, Desai S, Udwadia-Hegde A. Non-infantile variant of desmoplastic ganglioglioma: a report of 2 cases. Pediatr Radiol 2006 Jun;36(6):541-545.
- 7. Khaddage A, Chambonniere ML, Morrison AL, Allard D, Dumollard JM, Pasquier B, et al. Desmoplastic infantile ganglioglioma: a rare tumor with an unusual presentation. Ann Diagn Pathol 2004 Oct;8(5):280-283.
- 8. Geramizadeh B, Kamgarpour A, Moradi A. Desmoplastic infantile ganglioglioma: Report of a case and review of the literature. J Pediatr Neurosci 2010 Jan;5(1):42-44.
- 9. Trehan G, Bruge H, Vinchon M, Khalil C, Ruchoux MM, Dhellemmes P, et al. MR imaging in the diagnosis of desmoplastic infantile tumor: retrospective study of six cases. AJNR Am J Neuroradiol 2004 Jun-Jul;25(6):1028-1033.
- 10. Rypens F, Esteban MJ, Lellouch-Tubiana A, Bastien-Fagnou C, Renier D, Baraton J, et al. Desmoplastic supratentorial neuroepithelial tumours of childhood: imaging in 5 patients. Neuroradiology 1996 May;38(Suppl 1):S165-S168.
- 11. Fernandez C, Girard N, Paz Paredes A, Bouvier-Labit C, Lena G, Figarella-Branger D. The usefulness of MR imaging in the diagnosis of dysembryoplastic neuroepithelial tumor in children: a study of 14 cases. AJNR Am J Neuroradiol 2003 May;24(5):829-834.
- 12. Pommepuy I, Delage-Corre M, Moreau JJ, Labrousse F. A report of a desmoplastic ganglioglioma in a 12-year-old girl with review of the literature. J Neurooncol 2006 Feb;76(3):271-275.
- 13. Tamburrini G, Colosimo C Jr, Giangaspero F, Riccardi R, Di Rocco C. Desmoplastic infantile ganglioglioma. Childs Nerv Syst 2003 Jun;19(5-6):292-297.