Gestational trophoblastic disease (GTD) is a spectrum of disorders extending from premalignant conditions of hydatidiform mole, which can be further classified into partial (PHM) and complete (CHM) hydatidiform mole, to malignant conditions including invasive mole, choriocarcinoma, and placental site trophoblastic tumor (PSTT).1 This is based on the World Health Organization classification.2 Women diagnosed with gestational trophoblastic neoplasia (GTN) should be managed appropriately and treated with chemotherapy if required to reduce the risk of complications and metastasis.3 GTN, if left untreated, may lead to death.4 GTD occurs in 1 in 40 000 pregnancies and is commonly seen in Asia.2 The reported prevalence of GTD is 1 in 125 live births in Taiwan, 2 in 1000 pregnancies in South East Asia and Japan, 1 in 1000 pregnancies in Europe, and 1 in 1500 pregnancies in the USA.5 The reported incidence in Yemen is 1 in 164 pregnancies,6 1 in 318 in Iraq,7 and 1 in 314 pregnancies in Iran.8 The prevalence of hydatidiform mole in Brazil is 2.2%, South Africa 1.2 per 1000 deliveries and Nnewi, Southeast Nigeria is 0.3 per 1000 deliveries.9
Several associated risk factors increase a woman’s tendency to developing GTD. These include increased (older than 40 years) or decreased (under 20 years) maternal age, decreasing number of births, history of prior molar pregnancy or miscarriage, and oral contraceptives use.10 There is limited evidence between developing GTD and smoking habits, alcohol consumption, diet, socioeconomic status, and herbicide exposure.11–13
All patients with GTD should be followed-up closely with beta-human chorionic gonadotropin (βhCG) levels, and pregnancy should be avoided during that period.14 Women with PHM or CHM are usually cured by surgical intervention. However, as many as 1 in 5 women with CHM will have some persistent molar tissue, which has a tendency to develop into an invasive mole. In a few cases, it may develop to choriocarcinoma. Further treatment is required in either case.11,15
Using sonography, CHM in the first trimester appears as a uterine cavity filled with multiple sonolucent areas of varying size and shape (known as a snowstorm appearance) without the presence of fetal structures,16 and it may be associated with ovarian theca lutein cysts.17 PHM presents as an enlarged placenta with multicystic avascular sonolucent spaces (‘Swiss cheese’ appearance), and a fetus can be demonstrated by ultrasound.18
Histopathologically, CHM has a typical appearance of a voluminous mass of grape-like structures of chorionic villi, which are cystically dilated and swollen.18 For instance, CHM appears as circumferential trophoblast hyperplasia and swollen avascular villi.19 On the other hand, PHM, which is compatible with early embryogenesis with the formation of some triploid fetal parts, has some normal chorionic villi.18
Sultan Qaboos University Hospital (SQUH) is one of the largest tertiary centers in Oman and as far as we know GTD has not been studied in Oman. We sought to assess the prevalence and outcomes of GTD among pregnant women admitted at SQUH. Moreover, we aimed to determine the histological features, correlate them with sonographic findings, and estimate the minimum follow-up period required for βhCG levels to normalize.
Methods
We conducted a retrospective cohort study between November 2007 and October 2015 on all women diagnosed with GTD at SQUH. Cases were excluded from analysis for women lost to follow-up or with missing data. Ethical approval was obtained from the College of Medicine and Health Sciences Ethics Committee, Sultan Qaboos University (MREC no.1099).
The hospital database including hospital information system (electronic medical records), gynecology operating theater and histopathology laboratory registries were used to collect the following data: (1) maternal demographics and risk factors for GTD (age, gravidity, parity, gestational age of pregnancy, previous miscarriages, prior molar pregnancy, smoking and alcohol consumption, use of birth control pills, and family history of GTD); (2) histopathological features and types of GTD (PHM, CHM, invasive mole, choriocarcinoma, and PSTT) and the total number of deliveries during the study period; (3) information on the ultrasound features for each woman and the level of βhCG at the time of diagnosis and during follow-up periods; (4) details of the surgical intervention and complications; (5) postoperative use of chemotherapy.
We used the STATA (StataCorp. Stata Statistical Software: Release 13. College Station, TX) for data analysis. Descriptive statistics were used to describe the demographic characteristics, baseline clinical characteristics, histopathological, and sonographic features. To test the association between the GTD and its risk factors, we used the chi-square test. A p-value ≤ 0.050 was considered statistically significant.
Results
A total of 64 women diagnosed with GTD at SQUH were included in the study. The mean maternal age on admission was 31.0±7.5 years. The mean gravidity, parity, and abortion were 4.0±3.6, 2.0±3.2 and 1.0±1.0, respectively. The mean gestational age on admission was 11.0±3.2 weeks [Table 1]. Sixty women (93.8%) were diagnosed before 16 weeks gestation while only four women were diagnosed between 18 and 22 weeks gestation. None were diagnosed after 22 weeks.
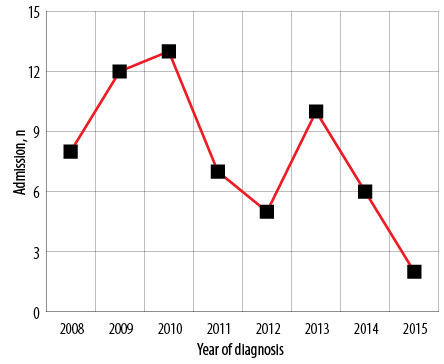
Figure 1: Prevalence of gestational trophoblastic disease in Sultan Qaboos University Hospital from January 2008 to October 2015.
One woman was diagnosed with GTD in 2007. However, the total number of GTD cases diagnosed in 2008, 2009, 2010, 2011, 2012, 2013, and 2014 was 8, 12, 13, 7, 5, 10, and 6, respectively. Only two women were diagnosed in 2015. Based on total number of deliveries at SQUH of 23 235 from 2008 to 2014, the prevalence of GTD in 2008 was 0.3% while in 2009, 2010, 2011, 2012, 2013, and 2014 were 0.5, 0.4, 0.2, 0.1, 0.3, and 0.1%, respectively [Figure 1]. The prevalence of GTD from 2008 to 2014 (seven years) was 0.3% equivalent to 1 in every 386 births.
Table 1: Maternal demographics of the study participants (n = 64).
|
Age at admission, years |
31.0 ± 7.5 (19–54) |
|
Gravidity, n |
4.0 ± 3.6 (1–17) |
|
Parity, n |
2.0 ± 3.2 (0–15) |
|
Abortion, n |
1.0 ± 1.0 (0–3) |
GTD was noted more in older and multiparous women, but this was not statistically significant (p = 0.285 and 0.399, respectively). There was no significant association noted between GTD and previous miscarriage or prior GTD (p = 0.446 and 0.140, respectively). None of the study participants reported smoking, drinking alcohol, use of oral contraceptive pills, or a family history of GTD.
Among the study sample, 28 (43.8%) women were diagnosed histopathologically to have CHM while PHM was seen in 35 (54.7%) women. An invasive mole was reported in one woman (1.6%). However, none of the women had PSTT or choriocarcinoma.
The majority of women diagnosed with CHM (89.3%, 25/28), showed snowstorm appearance (p = 0.005) [Table 2]. In women diagnosed with PHM, the features on ultrasound were the following: 12 (34.3%) had the same appearance of a missed miscarriage, and 11 (31.4%) had cystic placental changes with a fetal pole or an intrauterine gestational sac (p = 0.004 and 0.003, respectively). One woman with invasive mole was found to have a snowstorm appearance on scan. Moreover, two women with CHM and one with PHM had bilateral ovarian theca lutein cysts. The sonographic features were in correlation with the final histopathological diagnosis of all types of GTD, and this was statistically significant as mentioned above.
Table 2: Percentages of sonographic features in different types of GTD.
|
Snowstorm appearance |
5.7 |
89.3 |
100 |
0.005 |
|
Cystic placental changes with fetus or IUGS* |
31.4 |
0.0 |
0.0 |
0.004 |
|
Cystic placental changes without fetus or IUGS |
28.6 |
3.6 |
0.0 |
0.084 |
GTD: gestational trophoblastic disease; PHM: partial hydatidiform mole; CHM: complete hydatidiform mole; IM: invasive mole; IUGS: intrauterine gestational sac.
All women underwent suction evacuation and curettage under general anesthesia and syntocinon use. The mean estimated blood loss during the procedure was 156.0±136.0 mL. The maximum loss was 800 mL while the minimum loss was 100 mL. Among the 64 women with GTD, only four had severe bleeding (i.e., blood loss < 500 mL) which necessitated further uterotonic medications to avoid further complications. None of the women required a blood transfusion. There was no significant difference noted in estimated blood loss between PHM and CHM cases (p = 0.480).
In our study, among the 64 women, 41 were followed-up with βhCG until it reached the negative value (< 5 IU/L) after a mean follow-up period of 70 days. Twenty-three women were missed in the follow-up period as they were referred to a closer hospital.
Out of the 41 women whose data was reported completely, 22 had CHM. Their mean βhCG level at the time of diagnosis was 276 526.3 IU/L (range = 46860.0–1 155 527.0 IU/L). These women required a mean period of 64.0 days for their βhCG level to reach the negative value (range = 28–163 days). On the other hand, the mean βhCG levels of 18 women with PHM was 122 987.9 IU/L (range = 93.9–427 364.0 IU/L). A mean period of 62.0 days was required to reach the normal value of βhCG (range = 22–91 days). There was no significant difference between CHM and PHM cases in the time required for βhCG to normalize (p = 0.700).
The woman with invasive mole had a βhCG value of 284 263.0 IU/L at the time of diagnosis, and it took 328 days to reach the negative value. This woman was treated with surgical intervention and chemotherapy.
Fifty-seven (89.1%) of the 64 women diagnosed with GTD were treated completely by suction evacuation and did not require further treatment with chemotherapy while seven women (10.9%) had chemotherapy. Among those seven women, none had severe bleeding, which means that there was no significant association between severe bleeding and the need for chemotherapy (p = 0.374).
Among the 35 women diagnosed with PHM, only one required chemotherapy. Five of the 28 women with CHM required chemotherapy. There was a significant association between the GTD type and the need for chemotherapy (p = 0.013).
Discussion
This study included 64 women diagnosed with GTD. In the study period, the prevalence of GTD was 1 in 386 births. GTD types included 35 women with PHM, 28 with CHM and only one woman diagnosed with invasive mole. Comparing the prevalence of GTD between our study and some Asian countries, we noted a significant variation. In 2009, our study showed a higher prevalence of GTD, with one case diagnosed in every 201 births compared to a descriptive observational study conducted in Iraq that revealed the prevalence of GTD was 1 in every 318 births.7 In 2011, our findings showed that for every 475 births there was a single case diagnosed with GTD in contrast to a descriptive observational study conducted in Yemen in the same year, which reported a higher prevalence of one in every 164 births.6 Moreover, between 2012 and 2013, our results illustrated a single case of GTD diagnosed for every 453 births. The prevalence was higher in an Iranian study, with a rate of one in every 143 births.20 The most likely explanation for these variations between our study and some of the Asian countries is that this study was conducted only in a single tertiary institution in Oman while the previously mentioned studies were conducted country-wide. Moreover, these variations may be attributed to the smaller sample size in our study.
The majority of GTD cases in our study were diagnosed in women aged 26–39 years contrary to the findings published in other studies, which observed a higher risk of GTD among women under 20 years or older than 40 years old.10,17,21 Multiparous women constituted the majority of the cases as shown in a study from India.18 However, these results are in disagreement with the results published in many other studies, which reported that risk of GTD decreases with an increase in the number of births.12,13 Moreover, a study conducted in Mexico concluded that parity did not affect the risk of GTD.22 Unlike other published studies that demonstrated a significant association between several associated risk factors and the tendency to develop GTDs as prior miscarriage or GTD, family history of GTD, and use of oral contraceptives, our study was not able to show such an association.10–13,17,23 These variations could be attributed to the small sample size and that the study is retrospective in nature with the possibility of missing some information.
In our study, all women were treated by surgical intervention and seven (10.9%) required chemotherapy out of which five had CHM, one had PHM, and one had an invasive mole. Our study showed a significant association between the GTD type and chemotherapy treatment (p = 0.013) like other reported studies.2,14,24
Regarding the sonographic findings, 89.3% of CHM cases showed the typical snowstorm appearance picture on ultrasound. Furthermore, the false negative diagnosis of missed miscarriage was reported by ultrasound in 34.3% and 7.1% of PHM and CHM cases, retrospectively. The typical appearance of a PHM on ultrasound (cystic placental changes with a fetus or intrauterine gestational sac) was reported in 31.4% of cases. These ultrasound results highlight the importance of proper clinical assessment, looking into βhCG levels and the final histological results to reach the correct diagnosis and make the necessary follow-up.25,26 Ovarian luteal cysts are usually associated with CHMs due to the high levels of βhCG.14 These cysts were seen in two of CHM cases and one PHM case.
Surgical intervention needs a period of follow-up measuring βhCG levels to exclude the persistence or recurrence of the disease.16
According to our study, the estimated mean period for βhCG level to revert to the normal limit was 64 days for CHM and 62 days for PHM, and there was no significant difference between them. Our result is similar to other studies in which the mean period required to reach negative value was 56 days.14,27
Our study had a number of possible limitations particularly our use of a single center, small sample size, and the retrospective nature of the study, which may have contributed to missing data.
Conclusion
Our study highlights the importance of proper assessment and follow-up of the final histopathological diagnosis in women who undergo surgical evacuation to treat GTD. GTD, if not treated and followed-up appropriately, may lead to major complications including death. Furthermore, our results can be used as guidance for establishing programs to detect risk factors and plan management for women with GTD. Further prospective studies in Oman including multiple centers are recommended. Establishing a local database for GTD and GTN is needed.
Disclosure
The authors declared no conflict of interest. No funding was received for this study. The abstract was previously published as a supplement in 16th Biennial Meeting of the International Gynecologic Cancer Society.
references
- 1. Petrich S, Vaughan M. Gestational trophoblastic disease. New Zealand Gynaecologic Cancer Group Guidelines; 2014.
- 2. Seckl MJ, Sebire NJ, Fisher RA, Golfier F, Massuger L, Sessa C; ESMO Guidelines Working Group. Gestational trophoblastic disease: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2013 Oct;24(Suppl 6):vi39-vi50.
- 3. Shih IeM. Gestational trophoblastic neoplasia–pathogenesis and potential therapeutic targets. Lancet Oncol 2007 Jul;8(7):642-650.
- 4. Lurain JR. Gestational trophoblastic disease I: epidemiology, pathology, clinical presentation and diagnosis of gestational trophoblastic disease, and management of hydatidiform mole. Am J Obstet Gynecol 2010 Dec;203(6):531-539.
- 5. Garner EI, Goldstein DP, Feltmate CM, Berkowitz RS. Gestational trophoblastic disease. Clin Obstet Gynecol 2007 Mar;50(1):112-122.
- 6. Ahmed IAM. Gestational trophoblastic disease in Al-Thawra Hospital, Sana’a-Yemen. Yemeni journal for medical sciences 2013;7.
- 7. Al Alaf SK, Ibrahim D. Prevalence and clinical observations of gestational trophoblastic diseases in maternity teaching hospital in Erbil city. WSEAS Transactions on. Biology and Biomedicine 2010;7(3):190-199.
- 8. Javey H, Sajadi H. Hydatidiform mole in southern Iran: a statistical survey of 113 cases. Int J Gynaecol Obstet 1978;15(5):390-395.
- 9. Kitange B, Matovelo D, Konje E, Massinde A, Rambau P. Hydatidiform moles among patients with incomplete abortion in Mwanza City, North western Tanzania. Afr Health Sci 2015 Dec;15(4):1081-1086.
- 10. Steigrad SJ. Epidemiology of gestational trophoblastic diseases. Best Pract Res Clin Obstet Gynaecol 2003 Dec;17(6):837-847.
- 11. Gestational trophoblastic disease. American Cancer Society. [cited 2015 April 1]. Available from: http://www.cancer.org/cancer/gestationaltrophoblasticdisease/detailedguide/index.
- 12. Parazzini F, Mangili G, La Vecchia C, Negri E, Bocciolone L, Fasoli M. Risk factors for gestational trophoblastic disease: a separate analysis of complete and partial hydatidiform moles. Obstet Gynecol 1991 Dec;78(6):1039-1045.
- 13. Altieri A, Franceschi S, Ferlay J, Smith J, La Vecchia C. Epidemiology and aetiology of gestational trophoblastic diseases. Lancet Oncol 2003 Nov;4(11):670-678.
- 14. Seckl MJ, Sebire NJ, Berkowitz RS. Gestational trophoblastic disease. Lancet 2010 Aug;376(9742):717-729.
- 15. Seckl MJ, Fisher RA, Salerno G, Rees H, Paradinas FJ, Foskett M, et al. Choriocarcinoma and partial hydatidiform moles. Lancet 2000 Jul;356(9223):36-39.
- 16. Jauniaux E. Ultrasound diagnosis and follow-up of gestational trophoblastic disease. Ultrasound Obstet Gynecol 1998 May;11(5):367-377.
- 17. Sebire NJ, Foskett M, Fisher RA, Rees H, Seckl M, Newlands E. Risk of partial and complete hydatidiform molar pregnancy in relation to maternal age. BJOG 2002 Jan;109(1):99-102.
- 18. Kumar N, Saxena YK, Rathi AK, Chitra R, Kumar P. Host and risk factors for gestational trophoblastic disease: a hospital-based analysis from India. Med Sci Monit 2003 Oct;9(10):CR442-CR447.
- 19. Howat AJ, Beck S, Fox H, Harris SC, Hill AS, Nicholson CM, et al. Can histopathologists reliably diagnose molar pregnancy? J Clin Pathol 1993 Jul;46(7):599-602.
- 20. Almasi A, Almassinokiani F, Akbari P. Frequency of molar pregnancies in health care centers of tehran, iran. J Reprod Infertil 2014 Jul;15(3):157-160.
- 21. Loukovaara M, Pukkala E, Lehtovirta P, Leminen A. Epidemiology of hydatidiform mole in Finland, 1975 to 2001. Eur J Gynaecol Oncol 2005;26(2):207-208.
- 22. Moore EL. Gestational Trophoblastic disease. [cited 2009 May 2]. Available from: https://emedicine.medscape.com/article/279116.
- 23. Lorigan PC, Sharma S, Bright N, Coleman RE, Hancock BW. Characteristics of women with recurrent molar pregnancies. Gynecol Oncol 2000 Sep;78(3 Pt 1):288-292.
- 24. Gerulath A. Gestational trophoblastic disease. SOGC clinical practice guidelines 2002;114.
- 25. Sebire NJ, Rees H, Paradinas F, Seckl M, Newlands E. The diagnostic implications of routine ultrasound examination in histologically confirmed early molar pregnancies. Ultrasound Obstet Gynecol 2001 Dec;18(6):662-665.
- 26. Dhanda S, Ramani S, Thakur M. Gestational trophoblastic disease: a multimodality imaging approach with impact on diagnosis and management. Radiol Res Pract 2014;2014(S1):842751.
- 27. Tidy J, Hancock B. The management of gestational trophoblastic disease. Royal College of Obstetricians and Gynaecologists 2010;1(38):1-12.