The prognosis of chronic myeloid leukemia (CML) has been revolutionized after the introduction of the first tyrosine kinase inhibitor (TKI), imatinib in the early 2000s, followed by the second-generation TKIs dasatinib, nilotinib and bosutinib, and the third generation TKI ponatinib.1–5 Their effective and durable action has allowed many patients to enjoy a near-normal life and increased life expectancy.6
Transforming growth factor-β1 is a key regulator of immune homeostasis.7 Its upregulation signaling by BCR-ABL (BCR-ABL1 fusion gene) is one mechanism through which the transformation of hemopoietic progenitor cells is promoted.8 Programmed death-ligand 1 plays a critical role in T-cell activation, proliferation, and cytotoxic secretion,9 and decreased levels during TKI therapy are indicative of at least partial reversal of immune cell exhaustion.10
Discontinuation of TKI may cause TKI withdrawal syndrome, manifesting as musculoskeletal pain and/or flushing, due to a decrease in interleukin 10, which calls for carefully planned discontinuation.
Chronic viral hepatitis is a major global health problem, contributing significantly to the development of chronic liver disease, cirrhosis, and hepatocellular carcinoma.11 Hepatitis B virus (HBV) reactivation may occur in patients receiving TKIs, a potentially fatal complication, and regulatory authorities recommend testing CML patients for hepatitis before initiating TKI therapy.12–17
Studies have shown that chemotherapy can induce HBV reactivation in asymptomatic carriers of hepatitis B e antibody and hepatitis B surface antigen (HBsAg), but only rarely in individuals with resolved HBV infection.18 TKIs may have been associated with HBV reactivation (HBVr), as they tend to inhibit T-cell proliferation and activation, thereby impacting the immune response.19,20 Regarding hepatitis C virus (HCV) reactivation, however, Yazici did not find any significant evidence.21
Although the prevalence of HBV and HCV have been studied in Oman, there is a dearth of data on viral hepatitis in Omani CML patients.20,21 Therefore, this study assessed the prevalence of viral hepatitis in Omani CML patients treated with TKIs, and evaluated hepatitis reactivation outcomes and their possible association with CML molecular remission and survival.
Methods
This retrospective cohort study was conducted at Sultan Qaboos University Hospital (SQUH), Muscat. After obtaining ethical approval from medical research and ethics committee (Ref. MREC # 1667 dated 2 May 2018), records of patients with CML treated from 2006 to 2020 were retrieved from the hospital’s electronic medical records. We included all patients diagnosed with CML at SQUH since 2006 with available data on hepatitis B and C. Patient files with missing data or who failed to follow-up were excluded. Data collected included demographic information and clinical data including diagnostic details, spleen size, length of follow up, TKIs used, and outcomes. Laboratory data included baseline hemoglobin levels, white blood count, platelet count, blood chemistry, CML diagnostic markers, and viral hepatitis status.
Since 2011, monitoring of CML was performed using the GeneXpert system (Cepheid, Sunnyvale, CA, USA). HBVr was defined as any of the following: (a) a 2–3-fold increase in alanine aminotransferase above baseline; (b) HBsAg seroconversion; (c) newly detected HBV DNA; or (d) a ≥10-fold increase in HBV DNA level compared with the pre-immunosuppression baseline. All patients with positive hepatitis serology were monitored with regular polymerase chain reaction (PCR) (every 3–6 months) for HBV-DNA positivity.
Statistical analysis was performed using IBM SPSS Statistics (IBM Corp. Released 2015. IBM SPSS Statistics for Windows, Version 23.0. Armonk, NY: IBM Corp.). The prevalence of hepatitis infection and the proportion of patients meeting criteria for viral hepatitis reactivation after receiving TKIs were summarized as counts and percentages as appropriate. Survival analyses used dates of CML diagnosis, remission, relapse, last follow-up, and death from hospital records. Overall survival (OS) was defined as the time from diagnosis of CML to last follow-up or death. Progression-free survival was defined as the time from diagnosis to last follow-up, progression, relapse, or death. The Kaplan-Meier method was used to calculate survival curves. Survival comparisons were made using the log-rank test. A p-value of < 0.05 was considered statistically significant.
Results
The study included 98 Omani patients diagnosed with CML at SQUH from 2006 to 2020, with a mean age of 41.1 (range = 4–77) years [Table 1]. Females were in slight majority (54%). Sixty-eight (69.4%) patients received hydroxyurea before starting TKIs. Treatment with imatinib as first line was given to 93 (94.9%) patients; subsequently, 35 patients switched to imatinib, 20 to nilotinib, and 14 to dasatinib.
Table 1: Clinical, laboratory features, and viral hepatitis status in 98 CML patients on TKIs.
|
Mean age (range), years
|
41.1 (4–77)
|
|
Sex, n
|
|
|
Male
|
45
|
|
Female
|
53
|
|
Hematological parameters at presentation, mean (range)
|
|
Hb, g/dL
|
10.4 (4.1–14.9)
|
|
WBC, × 109/L
|
115.0 (2–600)
|
|
Platelets, ×109/L
|
456.0 (23–1710)
|
|
Spleen size in cm, mean (range)
|
14.8 (6–26)
|
|
Median follow up time
in months (IQR)
|
77 (2–24)
|
|
Alive, n (%)
|
89 (90.8)
|
|
Exposure to TKIs, n (%)
|
|
|
Imatinib
|
93 (94.9)
|
|
Dasatinib
|
23 (23.55)
|
|
Nilotinib
|
25 (25.5)
|
|
Exposure to ≥ 2 TKIs
|
48 (48.9)
|
|
Currently on CML therapy, n (%)
|
|
|
Imatinib
|
35 (39.3)
|
|
Dasatinib
|
14 (15.7)
|
|
Nilotinib
|
20 (22.5)
|
|
Others
|
4 (4.5)
|
|
TFR
|
9 (10.1)
|
|
Hepatitis B status, n (%)
|
|
|
HBV immune
|
45 (45.9)
|
|
HBV negative
|
77 (78.6)
|
|
HBV positive (including core)
|
16 (16.3)
|
|
HBsAg positive
|
6 (6.1)
|
|
HBsAg positive, positive HBV PCR
|
6 (6.1)
|
|
Hepatitis C status, n (%)
|
|
|
HCV serology positive
|
1 (1.1)
|
CML: chronic myeloid leukemia; TKI: tyrosine kinase inhibitors; Hb: hemoglobin; WBC: white blood cell; TFR: treatment free remission; HBV: hepatitis B virus; HBsAg: hepatitis B surface antigen; HCV: hepatitis C virus; PCR: polymerase chain reaction.
Kaplan-Meir plots illustrate the rates of progression-free survival and OS [Figures 1 and 2]. The mean OS rates of this cohort were excellent, with five-year and 10-year survival at 93.4% and 89.6%, respectively. There were 35 cases of interim CML relapse; all were salvaged with targeted therapeutic modalities including bone marrow transplantation. A total of 14 patients achieved treatment free remission (TFR) with persistent negative BCR-ABL with an RQ-PCR sensitivity of MR4.5 including five recipients of allogenic bone marrow transplantation. As per the follow-up records, two patients were receiving antiviral therapy with ponatinib/interferon. In this cohort, 11 (11.2%) patients were lost to follow-up, which included nine (9.2%) deaths.
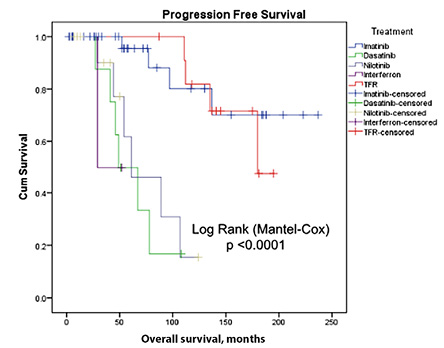 Figure 1: Kaplan-Meier plot for progression-free survival in the different categories of treatment modalities in patients with chronic myeloid leukemia (N = 98).
Figure 1: Kaplan-Meier plot for progression-free survival in the different categories of treatment modalities in patients with chronic myeloid leukemia (N = 98).
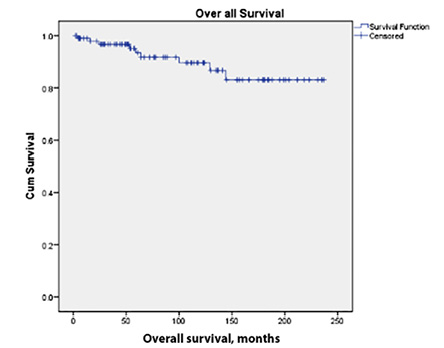 Figure 2: Kaplan-Meier plot for overall survival in chronic myeloid leukemia patients (N = 98).
Figure 2: Kaplan-Meier plot for overall survival in chronic myeloid leukemia patients (N = 98).
HBsAg was positive in six patients (6.1%; four males, two females) at baseline, all of whom were HBV PCR-positive [Table 2]. Anti-HB core (anti-HBc) was positive in 16 (16.3%) patients, including all the six patients with HBsAg positivity. All the six HBV PCR-positive patients were exposed to imatinib and other TKIs for median 58.0 (range = 4–24) months as shown in Table 2. Regarding antiviral therapy, Patient 3 was initially started on lamivudine and adefovir. At the last follow up, all six HBV PCR positive patients were on antiviral entecavir therapy [Table 2].
Table 2: Details of hepatitis B positivity and their therapy in CML patients on TKI (n = 6).
|
Year of diagnosis
|
2001
|
2006
|
2014
|
2016
|
2016
|
2017
|
|
Sex and age at diagnosis
|
15 M*
|
43 M*
|
28 M*
|
36 F
|
29 M
|
39 F
|
|
Hepatitis diagnosis at presentation
|
HBV
|
HBV
|
HBV
|
HBV
|
HBV
|
HBV
|
|
BCR-ABL transcript†
|
|
|
|
|
|
|
|
At diagnosis
|
NA
|
> 1
|
19
|
18
|
84
|
39
|
|
One year
|
NA
|
0.032
|
0.63
|
0.22
|
0.046
|
0.52
|
|
Currently
|
0.072
|
0.0002
|
0.015
|
0.081
|
0.031
|
0.004
|
|
Therapy for CML
|
|
|
|
|
|
|
|
Imatinib
|
✓
|
✓
|
✓
|
✓
|
✓
|
✓
|
|
Dasatinib
|
✓
|
✓
|
✓
|
|
|
|
|
Nilotinib
|
|
|
✓**
|
|
✓
|
✓
|
|
Ponatinib
|
✓
|
|
|
|
|
|
|
Interferon
|
|
|
|
|
|
✓
|
|
Antiviral therapy for hepatitis
|
|
|
|
|
|
|
|
Lamivudine
|
|
✓
|
|
|
|
|
|
Adefovir, dipivoxil
|
|
✓
|
|
|
|
|
|
Entecavir
|
✓
|
✓
|
✓
|
✓
|
✓
|
✓
|
|
Interferon, ribavirin
|
|
|
|
|
|
|
CML: chronic myeloid leukemia; TKI: tyrosine kinase inhibitor; HBV: hepatitis B virus; BCR-ABL: BCR-ABL1 fusion gene; NA: not available; ✓: treatment provided. †Values originally reported are transcribed here unchanged, despite differences in formats. *Patients 1–3 had complicated courses featured in more detail in Table 3. **Nilotinib was started because the patient was intolerant to dasatinib.
The one HCV RNA positive (genotype 1) patient in our cohort received imatinib concomitantly with interferon-based therapy and achieved remission from CML and hepatitis C for over 10 years and on last follow-up, she was off-therapy for both conditions.
Three patients [Patients 1–3 in Table 2] experienced particularly complicated courses. Despite this, all three achieved good outcomes, attributable to carefully combined anti-viral therapy and TKIs. Their details are presented in Table 3.
Patient 1 was an adolescent boy diagnosed with CML and HBV PCR positivity, with obesity and fatty liver as comorbidities. Due to resistance to imatinib and lack of compliance, TKI was switched to dasatinib. After eight years, upon confirmation of an atypical mutation, the TKI was changed to ponatinib. Currently, he has a significantly reduced CML transcript level. Sharply increased HBV DNA during treatment was managed with antiviral entecavir, resulting in undetectable HBV PCR, with normal liver enzyme levels [Tables 2 and 3].
Table 3: Treatment course of selected patients with complicated clinical courses (n = 3).
|
Patient 1. Male; 15 y
|
|
04/2009
|
12%
|
34/23
|
< 50
|
|
Imatinib
|
Obesity, fatty liver
|
|
01/2011
|
65.5%
|
64/244
|
20
|
|
Imatinib
|
|
11/2011
|
86.5%
|
74/44
|
23
|
|
Dasatinib
|
|
02/2012
|
24%
|
66/28
|
892
|
|
Dasatinib
|
|
09/2013
|
15%
|
38/28
|
2052
|
|
Dasatinib
|
|
06/2014
|
8.5%
|
44/29
|
898
|
|
Dasatinib
|
|
06/2015
|
25%
|
29/24
|
79
|
|
Dasatinib
|
|
06/2018
|
16
|
46/27
|
285 981
|
|
Dasatinib
|
|
09/2018
|
13
|
24/24
|
1328
|
|
Ponatinib
|
|
09/2019
|
0.27
|
19/17
|
13 734
|
|
Ponatinib
|
|
05/2020
|
0.019
|
26/21
|
undetectable
|
Entecavir
|
Ponatinib
|
|
Patient 2. Male; 43 y
|
|
12/2006
|
> 1
|
55/37
|
Positive
|
|
Imatinib
|
|
|
09/2007
|
0.1
|
44/25
|
Positive
|
Lamivudine
|
Imatinib
|
|
04/2008
|
0.032
|
42/26
|
Positive
|
Lamivudine
|
Imatinib
|
|
06/2010
|
Pos*
|
43/29
|
Negative
|
Adefovir
|
Imatinib
|
|
07/2011
|
0.16
|
55/33
|
Negative
|
Adefovir
|
Imatinib
|
|
07/2012
|
0.15
|
39/25
|
Negative
|
Adefovir
|
Imatinib
|
|
12/2015
|
0.008
|
45/27
|
Positive
|
Entecavir
|
Imatinib
|
|
03/2016
|
0.003
|
37/24
|
Negative
|
Entecavir
|
Imatinib
|
|
05/2017
|
0.001
|
137/81
|
Negative
|
Entecavir
|
Dasatinib
|
|
06/2018
|
0.001
|
20/22
|
Negative
|
Entecavir
|
Dasatinib
|
|
06/2019
|
0.0001
|
21/22
|
Negative
|
Entecavir
|
Dasatinib
|
|
03/2020
|
0.0002
|
19/19
|
Negative
|
Entecavir
|
Dasatinib
|
|
Patient 3. Male; 28 y
|
|
12/2014
|
19
|
11/17
|
28 805
|
Entecavir
|
Imatinib
|
Severe diarrhea
|
|
12/2015
|
0.41
|
14/21
|
< 20
|
Entecavir
|
Imatinib
|
|
10/2016
|
0.19
|
14/18
|
Negative
|
Entecavir
|
Imatinib
|
|
02/2017
|
0.100
|
14/21
|
Negative
|
Entecavir
|
Dasatinib
|
|
12/2018
|
0.06
|
26/18
|
Negative
|
Entecavir
|
Nilotinib
|
|
03/2019
|
0.032
|
26/19
|
Negative
|
Entecavir
|
Nilotinib
|
†Values originally reported are mentioned here unchanged, despite differences in formats. Pos: positive but not quantifiable; ALT: alanine transaminase; AST: aspartate transaminase; HBV: hepatitis B virus; TKI: tyrosine kinase inhibitor; CML: chronic myeloid leukemia.
Patient 2 presented with CML and hepatitis. He was commenced on imatinib as TKI and lamivudine as antiviral. He quickly reached deep molecular remission with undetectable BCR-ABL1 transcripts at sensitivity of > MR4. Thereafter, he became HBV PCR positive, albeit with a low viral transcript levels and mildly elevated transaminases. Though the switch to adefovir initially resulted in negative HBV PCR, the condition returned with mild transaminitis, and eventually resolved with entecavir therapy. However, CML relapsed after ten years on imatinib, necessitating a switch to dasatinib which restored BCR-ABL negativity with an RQ-PCR sensitivity of MR4.5 [Tables 2 and 3].
Patient 3 was initially treated with imatinib for CML and entecavir for HBV. Although the virus cleared with long-term remission, the CML did not respond to imatinib. Switching to dasatinib achieved a deep molecular remission, but he developed a severe bloody diarrhea. This was resolved by switching to nilotinib, which continued to provide a good molecular response to CML. At the last available follow-up he remained negative for HBV PCR, with normal liver enzymes [Tables 2 and 3].
Discussion
Viral hepatitis reactivation is a recognized risk for patients receiving anticancer treatments, including TKIs used in chronic myeloid leukemia.6,11–15 Due to limited data from Oman, this retrospective study assessed the prevalence, reactivation, and outcomes of viral hepatitis in 98 Omani patients with CML.
Our patients’ mean age of 41.1 years is similar to other regional studies, and much lower than in Western cohorts.2,22,23 Female patients were in the majority unlike in Western reports.2,23
Baseline hemoglobin, white blood cell, and platelet were similar to those previously reported from our region.23–25 Imatinib was more frequently used as first-line treatment. About 50% of patients continued on imatinib in the long term, including 10.1% who discontinued this drug after a successful TFR attempt.26 Nilotinib and dasatinib were used in fewer cases. The overall 5-year and 10-year survival of this CML cohort is excellent, reaching 93.4% and 89.6%, respectively, similar to rates reported internationally.2 Death rate was 9.2%, but none was attributable to CML relapse. All 35 relapsed patients survived either by changing TKI (n = 30) or via bone marrow transplantation (n = 5).
Oman has an intermediate prevalence of HBV carriers according to World Health Organization criteria; the prevalence of HBV infection is 5.8%,27 whereas that of HCV is 0.41%.28 The prevalence of HBV and HCV in CML patients in this study was higher than in the general population.
HBV reactivation in patients who receive immunosuppressive agents is defined as a rise in transaminases about three times the upper limit of normal, with an abrupt increase in the HBV DNA, and a potentially fatal complication.14–18 Depending on the immunosuppressive agent used, the risk of reactivation is classified as low, intermediate, and high.29 Although TKIs are not believed to be immunosuppressant, they are identified as a moderate risk for HBV reactivation, with a few reported cases associated with imatinib and nilotinib treatments.13–17 Imatinib was shown in vitro to inhibit T-cell activation, while nilotinib is known to inhibit Src-family kinase lymphocyte-specific protein tyrosine kinase and interfere with T-cell proliferation and function.19 It has been suggested that transforming growth factor-β1 and programmed death-ligand 1 may be responsible for T-cell activation, proliferation, and cytotoxic secretion,8 and their decreased levels during TKI therapy suggest at least partial reversal of immune cell exhaustion.10
To date, HBVr has not been reported in patients treated with dasatinib, apart from a single case in Japan involving low-dose therapy in a patient with previously resolved HBV infection.11 All reported cases of HBVr occurred in HBsAg-positive carriers.13
In this cohort, 16 patients were anti-HBc positive at baseline. Among them, six patients with HBsAg positivity and detectable HBV DNA had varying viral loads. Before initiating TKIs, all six received nucleoside analogue (NA) therapy [Table 2]. Despite this, no evidence of HBV reactivation has been detected so far. Guidelines for treating patients with hepatitis surface antigen/DNA positivity recommend indefinite therapy with either interferon based agents or NA inhibitors.30–32 Entecavir is a potent NA inhibitor with lower risk of resistance.33 Accordingly, three of our six HBS-Ag positive patients were given entecavir, with consistent response and absence of viral DNA including those who received both imatinib and nilotinib.
In a Taiwanese study by Chen et al,16 HBVr occurred not only in HBsAg-positive patients but also in HBsAg-negative patients with anti-HBc antibody positivity and/or anti-hepatitis B surface antibody positivity. In our cohort, 16 patients were anti-HBc positive; however, 10 had negative HBV DNA PCR, and none developed HBVr despite exposure to both first and second generation TKIs. Patients with HBV infection (defined as detectable HBV DNA in the absence of HBsAg) may also have HBVr. In such cases HBVr is defined as either seroconversion of HBsAg or a 10-fold (1-log) increase above the lower limit of detectable HBsAg and HBV DNA, whose levels were previously undetectable. Accordingly, in patients with detectable baseline HBV DNA, a 1-log increase above baseline was considered indicative of HBVr.
In keeping with the current recommendations for patients who are at risk of HBVr,23 all our patients who were hepatitis B-positive received entecavir or adefovir for prophylaxis and treatment, except for one patient who was initially on lamivudine and shifted to entecavir following progression. All six patients became HBV PCR negative, with normal liver enzymes. We therefore recommend that patients with hepatitis receiving TKIs should have regular PCR testing (every 3–6 months) for HBV DNA positivity, and/or HBsAg seroconversion to detect any recurrence and treat it immediately. It is important to note that four of our patients with hepatitis B failed first-generation TKI therapy and needed second-generation (though not third-generation) TKIs. Though this gives us a hint of possible potential negative impact of hepatitis B infection on response to TKI therapy, the numbers are too small for statistical significance. Further, as the median time to HBVr is approximately 9–10 months (range = 1–69 months) after initiation of TKI therapy, current evidence supports starting antiviral prophylaxis at the initiation of TKI treatment in HBsAg-positive patients and continuing it for at least two years.23
No reports of hepatitis C reactivation while on imatinib therapy could be found in the literature.15 In the present cohort there was only one patient with HCV RNA positivity. She was treated with imatinib with concomitant interferon-alpha-based therapy leading to deep and persistent molecular remission with a negative BCR-ABL transcript, enabling cessation of both medications. Although this patient needed intermittent granulocyte colony stimulating factor to deliver both therapies on time, it is possible to successfully combine both anti-HCV and TKI therapy. This also highlights the role of interferon combination with TKIs in CML patients.34
The main limitation of this study is its retrospective design, which affected data quality and availability. This was a single-center study with a relatively small sample, and the cross-sectional follow-up was not sufficiently long for some cases. In addition, certain technologies, such as the GeneXpert machine for monitoring CML, were introduced later and may have had a minor impact on data comparability between earlier and later cases. Furthermore, BCR-ABL transcript data were reported in different formats over time, which may affect the interpretation of Tables 2 and 3. Therefore, any causal conclusions regarding the relationship between hepatitis B infection and CML progression, and the impact of CML therapy in triggering previously resolved HBV PCR positivity should be interpreted with caution.
Conclusion
Our study showed that patients with CML patients in this cohort were younger and predominantly female, with higher HBV and HCV rates compared with the general Omani population. Among the six patients with HBV infection, no evidence of viral reactivation was observed despite exposure to all three generations of TKIs. Antiviral prophylaxis appeared to prevent HBVr, as all HBV-infected patients maintained undetectable HBV DNA by PCR and normal liver enzymes during follow-up. Only one patient had HCV infection, which responded well to interferon therapy; the patient subsequently achieved deep molecular remission and TFR.
Disclosure
The authors declare no conflicts of interest. No funding was received for this study.
Acknowledgements
We wish to thank the SQUH administration for permitting the use of hospital resources for this study.
references
- 1. Goldman JM, Melo JV. Chronic myeloid leukemia–advances in biology and new approaches to treatment. N Engl J Med 2003 Oct;349(15):1451-1464.
- 2. Mughal TI, Radich JP, Deininger MW, Apperley JF, Hughes TP, Harrison CJ, et al. Chronic myeloid leukemia: reminiscences and dreams. Haematologica 2016 May;101(5):541-558.
- 3. Hochhaus A, Larson RA, Guilhot F, Radich JP, Branford S, Hughes TP, et al; IRIS Investigators. IRIS Investigators. Long-term outcomes of imatinib treatment for chronic myeloid leukemia. N Engl J Med 2017 Mar;376(10):917-927.
- 4. Cortes JE, Saglio G, Kantarjian HM, Baccarani M, Mayer J, Boqué C, et al. Final 5-year study results of DASISION: the dasatinib versus imatinib study in treatment-naive chronic myeloid leukemia patients’ trial. J Clin Oncol 2016 Jul;34(20):2333-2340.
- 5. Hochhaus A, Saglio G, Hughes TP, Larson RA, Kim D-W, Issaragrisil S, et al. Long-term benefits and risks of frontline nilotinib vs imatinib for chronic myeloid leukemia in chronic phase: 5-year update of the randomized ENESTnd trial. Leukemia 2016 May;30(5):1044-1054.
- 6. Bower H, Björkholm M, Dickman PW, Höglund M, Lambert PC, Andersson TM. Life expectancy of patients with chronic myeloid leukemia approaches the life expectancy of the general population. J Clin Oncol 2016 Aug;34(24):2851-2857.
- 7. Abdel Hammed MR, Ahmed YA, Adam EN, Bakry R, Elnaggar MG. sVCAM-1, and TGFβ1 in chronic phase, chronic myeloid leukemia patients treated with tyrosine kinase inhibitors. Egypt J Immunol 2022 Oct;29(4):163-173.
- 8. Møller GM, Frost V, Melo JV, Chantry A. Upregulation of the TGFbeta signalling pathway by Bcr-Abl: implications for haemopoietic cell growth and chronic myeloid leukaemia. FEBS Lett 2007 Apr;581(7):1329-1334.
- 9. Dufva O, El-Missiry MM, hteenma kiS. Mustjoki O. Expression of immune checkpoint molecules in chronic myeloid leukemia at diagnosis and during tyrosine kinase inhibitor therapy. November 2015, Annals of Oncology 26 (suppl 8): viii5–viii14.
- 10. Johnson-Ansah HA, Briant A, Charbonnier A, Cambier N, Damaj GL, Lenain P, et al. Interleukin 10 (IL10) and IL15 significantly decreased in chronic myeloid leukemia patients presenting with tyrosine kinase inhibitor (TKI) withdrawal syndrome (TWS). Results from the Prospective, Explorative and Multicenter « Kiwis » Study Designed for TWS. Blood 2023 Nov;142:6335 .
- 11. Lavanchy D. Chronic viral hepatitis as a public health issue in the world. Best Pract Res Clin Gastroenterol 2008;22(6):991-1008.
- 12. Ando T, Kojima K, Isoda H, Eguchi Y, Honda T, Ishigami M, et al. Reactivation of resolved infection with the hepatitis B virus immune escape mutant G145R during dasatinib treatment for chronic myeloid leukemia. Int J Hematol 2015 Sep;102(3):379-382.
- 13. Chang CS, Tsai CY, Yan SL. Hepatitis B reactivation in patients receiving targeted therapies. Hematology 2017 Dec;22(10):592-598.
- 14. Ikeda K, Shiga Y, Takahashi A, Kai T, Kimura H, Takeyama K, et al. Fatal hepatitis B virus reactivation in a chronic myeloid leukemia patient during imatinib mesylate treatment. Leuk Lymphoma 2006 Jan;47(1):155-157.
- 15. Chen CY, Tien FM, Cheng A, Huang SY, Chou WC, Yao M, et al. Hepatitis B reactivation among 1962 patients with hematological malignancy in Taiwan. BMC Gastroenterol 2018 Jan;18(1):6.
- 16. Wang YD, Cui GH, Li M, Gowrea B, Xia J, Hu Y. Hepatitis B virus reactivation in a chronic myeloid leukemia patient treated with imatinib mesylate. Chin Med J (Engl) 2012 Jul;125(14):2636-2637.
- 17. Lai GM, Yan SL, Chang CS, Tsai CY. Hepatitis B reactivation in chronic myeloid leukemia patients receiving tyrosine kinase inhibitor. World J Gastroenterol 2013 Feb;19(8):1318-1321.
- 18. Davison SA, Strasser SI. Ordering and interpreting hepatitis B serology. BMJ 2014 Apr;348:g2522.
- 19. Seggewiss R, Loré K, Greiner E, Magnusson MK, Price DA, Douek DC, et al. Imatinib inhibits T-cell receptor-mediated T-cell proliferation and activation in a dose-dependent manner. Blood 2005 Mar;105(6):2473-2479.
- 20. Mohty M, Jourdan E, Mami NB, Vey N, Damaj G, Blaise D, et al. Imatinib and plasmacytoid dendritic cell function in patients with chronic myeloid leukemia. Blood 2004 Jun;103(12):4666-4668.
- 21. Yazici O, Sendur MA, Aksoy S. Hepatitis C virus reactivation in cancer patients in the era of targeted therapies. World J Gastroenterol 2014 Jun;20(22):6716-6724.
- 22. Khaled SA, Nabih O, Abdel Aziz NM, Mahran DG. Myeloid leukemias: a glance at middle eastern centers. J Blood Med 2019 Dec;10:425-433.
- 23. Bawazir A, Al-Zamel N, Amen A, Akiel MA, Alhawiti NM, Alshehri A. The burden of leukemia in the Kingdom of Saudi Arabia: 15 years period (1999-2013). BMC Cancer 2019 Jul;19(1):703.
- 24. Radivoyevitch T, Jankovic GM, Tiu RV, Saunthararajah Y, Jackson RC, Hlatky LR, et al. Sex differences in the incidence of chronic myeloid leukemia. Radiat Environ Biophys 2014 Mar;53(1):55-63.
- 25. El-Ghammaz AM, Hamza MT, Said RM, Moussa MM, Eissa AM, Azzazi MO. Impact of additional chromosomal aberrations present at diagnosis on outcome of adolescent and young adult chronic myeloid leukemia patients: a single center experience. Indian J Hematol Blood Transfus 2019 Oct;35(4):683-691.
- 26. Pophali PA, Patnaik MM. The role of new tyrosine kinase inhibitors in chronic myeloid leukemia. Cancer J 2016;22(1):40-50.
- 27. Al Baqlani SA, Sy BT, Ratsch BA, Al Naamani K, Al Awaidy S, Busaidy SA, et al. Molecular epidemiology and genotyping of hepatitis B virus of HBsAg-positive patients in Oman. PLoS One 2014 May;9(5):e97759.
- 28. Mohamoud YA, Riome S, Abu-Raddad LJ. Epidemiology of hepatitis C virus in the Arabian Gulf countries: Systematic review and meta-analysis of prevalence. Int J Infect Dis 2016 May;46:116-125.
- 29. Loomba R, Liang TJ. Hepatitis B reactivation associated with immune suppressive and biological modifier therapies: current concepts, management strategies, and future directions. Gastroenterology 2017 May;152(6):1297-1309.
- 30. Liang R, Lau GK, Kwong YL. Chemotherapy and bone marrow transplantation for cancer patients who are also chronic hepatitis B carriers: a review of the problem. J Clin Oncol 1999 Jan;17(1):394-398.
- 31. European Association For The Study Of The Liver. EASL clinical practice guidelines: Management of chronic hepatitis B virus infection. J Hepatol 2012 Jul;57(1):167-185.
- 32. Chang TT, Lai CL, Kew Yoon S, Lee SS, Coelho HS, Carrilho FJ, et al. Entecavir treatment for up to 5 years in patients with hepatitis B e antigen-positive chronic hepatitis B. Hepatology 2010 Feb;51(2):422-430.
- 33. Fidan S, Fidan E, Karaman E, Erkut M, Cosar AM, Hepatitis B. Hepatitis B Reactivation in HBsAg-negative Anti-HBc-positive patients receiving cytotoxic chemotherapy for solid tumors. Acta Gastroenterol Belg 2020;83(3):426-431.
- 34. Talpaz M, Hehlmann R, Quintás-Cardama A, Mercer J, Cortes J. Re-emergence of interferon-α in the treatment of chronic myeloid leukemia. Leukemia 2013 Apr;27(4):803-812.