|
Abstract
We report here four cases of genital ulcers that developed after the administration of all-trans retinoic acid (ATRA) for the treatment of acute promyelocytic leukemia (APL). Between October 2007 and March 2010, three males and one female (age range 19-35 years) were identified to have genital ulcers after being prescribed all-trans retinoic acid (ATRA) as a part of chemotherapy for APL. This is the first series of cases describing genital ulcers, as a unique and rare complication of ATRA used for treatment of APL in these patients, with no other cause identified. Following temporary cessation of ATRA for a few days in these three cases, improvement of the ulcers was noted.
Keywords: Acute promyelocytic leukaemia; All-trans retinoic acid; Fever; Scrotal ulcers; ATRA.
Introduction
Acute promyelocytic leukemia (APL) is a homogeneous subgroup of acute myeloid leukemia (AML) characterized by the presence of (15;17) translocation causing PML/RARα fusion protein. All-trans retinoic acid (ATRA) has been shown to improve the outcome in patients with APL. It induces a complete remission (CR) in approximately 90% of patients when combined with chemotherapy.1
ATRA induces the differentiation of leukemic cells into phenotypically mature myeloid cells. Addition of ATRA to cytotoxic chemotherapy has been shown to improve the outcome in patients with APL.1-3 Adverse effects associated with ATRA include dryness of the skin, xerostomia, cheilitis, headache, bone pain, liver dysfunction, and pseudotumor cerebri.4 A more serious side effect is the ATRA syndrome, which consists of high fever, respiratory distress, and transient pulmonary infiltrates with leucocytosis.5,6
Unusual scrotal rashes, including exfoliative dermatitis, Sweet’s syndrome, and Fournier's gangrene have rarely been reported.7-9 The development of genital ulcer is a rare and unique complication of ATRA and has been rarely described.10-13 Here, we describe this rare complication in four patients with APL which is the largest case series reported in literature.
First Case
A 30-year-old man was admitted to our hospital because of diminishing vision in right eye, difficulty in swallowing and pain in throat with some blood tinged sputum for one week in February 2010. Ophthalmological examination revealed retinal hemorrhages. The rest of his examination was, otherwise, normal. Investigations revealed APL (see Table 1 for other investigations), and he was started with daily oral administration of ATRA 45 mg/m2 and intravenous idarubicin (12 mg/m2/day × 4 days). No ATRA syndrome was observed. On day 16 of his chemotherapy, he started complaining of ulcer on the tongue. There were no other complaints and the patient remained afebrile.
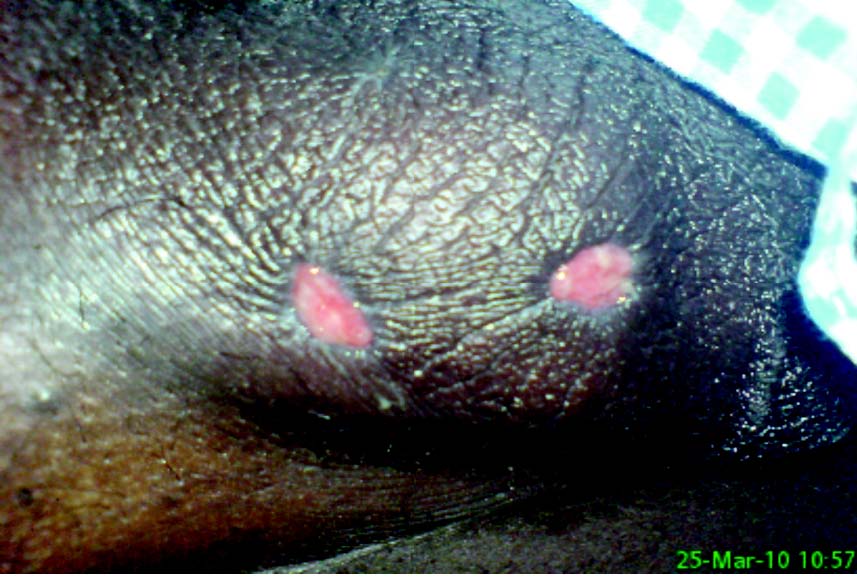
Figure 1: Showing well-circumscribed two scrotal ulcers (case 1)
He was started on antiviral and antifungal medications. The ulcer healed on the 20th day of chemotherapy, but he became febrile the next day. In spite of the administration of antibiotics and anti-fungal agent, fever persisted. Blood cultures were negative and the origin of the fever remained unclear. Painful scrotal vesicles appeared on day 22; the number of vesicles increased gradually and eventually converged and converted into two ulcers, (Fig. 1). ATRA was discontinued on day 26 after confirming morphological remission in the bone marrow. Ulcer swab was sent for culture, but it was negative for bacterial or viral pathogens. He defervesced over a few days, and the scrotal ulcers started improving on the 3rd day of stopping ATRA and took 9 weeks for complete healing. He was later re-challenged with ATRA with no recurrence of his genital ulcers.
Second Case
An 18-year-old male patient presented with recurrent history of fever and sweating, followed by left upper abdominal quadrant pain. Clinically, he had petechial rashes on upper trunk. There was no lymphadenopathy or splenomegaly. His initial investigations are shown in Table 1. He was diagnosed to have APL. After molecular confirmation, he was started with ATRA (45 mg/m2) and idarubicin (15 mg/m2/day for 3 days). Two days later, he developed oxygen desaturation and his chest X-ray revealed airspace disease bilaterally. ATRA syndrome was suspected and therefore ATRA was stopped for 5 days and started on dexamethasone 4 mg twice daily. He was transferred and stayed in the intensive care unit for 5 days. About 2 weeks after starting chemotherapy, he developed fever followed by right-sided scrotal swelling with small superficial ulcers on the same side. Skin swabs were negative for viral and bacterial cultures. In view of the fever and scrotal swelling, he was treated with broad spectrum antibiotics in addition to antiviral and antifungal.
He was reviewed by both surgeons and urologist for his ulcers and was suspected to be ATRA induced. ATRA was stopped for 6 days. His fever settled and the scrotal ulcer started to heal. Thereafter, the ATRA was restarted. His ulcers continued to heal and were completely healed by 4 weeks. Furthermore, he achieved a morphological complete remission (CR) on repeat bone marrow examination by day 28. On his subsequent consolidation chemotherapies, he did not develop recurrence of scrotal ulcers despite being on ATRA again.
Third Case
A 23-year-old lady, previously healthy, presented with bruises in the lower limbs, epistaxis, and mild hemoptysis associated with fever. At presentation, she had vaginal bleeding as part of her menstrual cycle. Examination revealed pallor with bruises in the thigh and the arms. No visceromegaly or lymphadenopathy was evident. On investigations, she was found to have APL (see Table 1). She was given ATRA (45 mg/m2) and idarubicin (12 mg/m2) from the 2nd day for 4 days. No ATRA syndrome was observed. On the 16th day after commencement of ATRA, she developed perineal lesion with labial swelling and two superficial ulcers with fever. She was reviewed by gynecologists as well. Cultures and swab were taken with initiation of antibiotics, antiviral and antifungal. All the culture results were negative for viruses and bacteria. On the 22nd day, ulcers started drying up with resolution of fever. ATRA was continued. The patient achieved morphological CR on bone marrow aspiration done on day 36. The ulcer healed over a period of 8 weeks.
Fourth Case
A 22-year-old male patient was admitted with fever, throat pain, myalgia and mild cough. He had occasional epistaxis. His examination revealed ecchymoses at venipuncture sites. Throat was congested with left tonsillar hypertrophy. No lymphadenopathy or visceromegaly. On investigations, he was confirmed to have APL (see Table 1). Treatment with ATRA at 45 gm/m2 was started and daunorubicin was added on the 3rd day after starting ATRA.
After 20 days of starting chemotherapy, he developed fever and scrotal edema with multiple scrotal ulcers. He was started on antibiotics with no improvement. He was febrile with conjunctival hemorrhages. The scrotal examination showed multiple ulcers with swelling and tenderness. ATRA was held after the suspicion that the ulcers were induced by it. He was reviewed by the surgeons and his cultures and genital swabs did not reveal infectious etiology. His ulcers started to heal gradually with complete resolution after 8 weeks. He did not develop the genital ulcers despite the reintroduction of ATRA during consolidation chemotherapy.
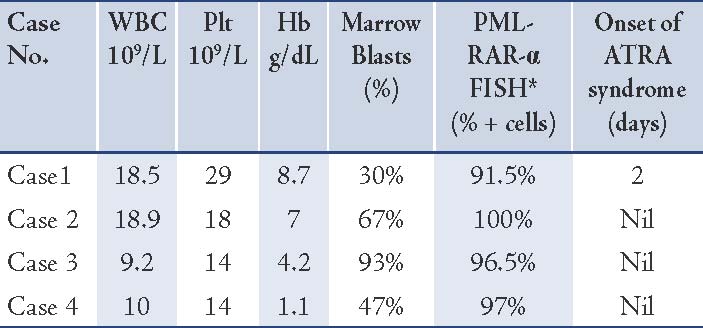
Table 1: Basic characteristics of the cases including baseline complete blood count, % marrow blasts, % of PML-RAR alpha positive cells, development and timing of ATRA syndrome and onset of genital ulcers after starting ATRA:
Discussion
The introduction of ATRA has remarkably improved the treatment and survival of patients diagnosed with APL. By inducing maturation and subsequent apoptosis of APL cells, ATRA therapy combined with chemotherapy has led to high percentage of complete remissions. Further induction, consolidation, and maintenance chemotherapy is needed to sustain remissions.14
Four cases with a rare complication of multiple ulcers localized in the scrotum and/or vulva, accompanied by high fever, occurring during the induction therapy with ATRA are presented here. Fever promptly improved with discontinuation of ATRA in three cases followed by healing of the ulcers in all cases. (See Tables 1 and 2)
ATRA-induced scrotal ulceration was first reported by Sun et al.15 in 1993 in a Chinese patient with severe exfoliative dermatitis and ulceration of the scrotum. The ulcers are usually observed between the 9th and 22nd days of ATRA treatment and they have been reported to resolve over 3 weeks to 3 months with the use of local or intravenous corticosteroids.8 In the four patients, the range of onset of the ulcers was 16 to 22 days. Although the cause of ATRA-induced scrotal ulcerations remains obscure, there are several proposed explanations. The ulcers may result from the release of various cytokines like tumour necrosis factor-α, interleukin IL-1, IL-6, and IL-8 that stimulate peripheral leukocytes and can lead to ulcerations.7 Moreover, ATRA is reported to directly activate leukocytes by producing oxygen radicals like superoxide, which can potentially cause tissue damage.16 In other reported cases, the scrotal ulcerations occurred when the white blood cell count was recovering.16
Considering the major benefit of ATRA therapy, it is reasonable to consider continuing ATRA therapy even when scrotal ulceration develops. In addition, steroids and antibiotics might be used to prevent secondary infection and to manage ulceration respectively.10 Topical corticosteroids and antibiotic ointments to prevent superimposed infection have been used to manage the ulceration and might be beneficial. We have chosen to temporarily discontinue ATRA in 3 patients given the progressive nature and the risk of developing Fournier’s gangrene. The decision to stop ATRA temporarily in these patients should be considered on each individual case and should be judged clinically especially if other interventions like local or occasionally systemic steroids do not succeed in controlling the ATRA related ulcers.
Given their anatomic location and painless nature, some patients may be hesitant reporting these ulcerations. Therefore, the incidence of these lesions in patients receiving ATRA therapy is probably underestimated. Despite the fact that treatment is most often continued through this cutaneous side effect, it is important for physicians to recognize these ulcers and distinguish them from cutaneous infections or the skin lesions associated with Sweet's syndrome.17 Also in all the 4 patients, anthracyclines as per standard protocol was given and whether these ulcers are related to the chemotherapy or not is not clear.
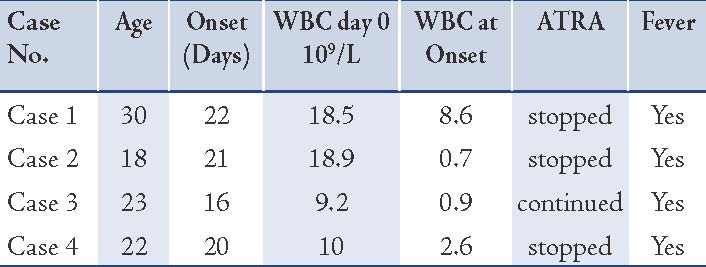
Table 2: Genital ulcer during treatment of APL with ATRA.
Conclusion
We report these cases to highlight its rarity, the importance of early recognition of genital ulcers following ATRA therapy and importance of differentiation from other causes like infection. Clinicians should be aware of this distressing complication, especially as ATRA is a very valuable and an integral part of the treatment of APL.
Acknowledgements
The authors reported no conflict of interest and no funding was received for this work.
References
1. Adès L, Sanz MA, Chevret S, Montesinos P, Chevallier P, Raffoux E, et al. Treatment of newly diagnosed acute promyelocytic leukemia (APL): a comparison of French-Belgian-Swiss and PETHEMA results. Blood 2008 Feb;111(3):1078-1084.
2. Jácomo RH, Melo RA, Souto FR, de Mattos ER, de Oliveira CT, Fagundes EM, et al. Clinical features and outcomes of 134 Brazilians with acute promyelocytic leukemia who received ATRA and anthracyclines. Haematologica 2007 Oct;92(10):1431-1432.
3. da Costa Moraes CA, Trompieri NM, Cavalcante Felix FH. Pediatric acute promyelocytic leukemia: all-transretinoic acid therapy in a Brazilian pediatric hospital. J Pediatr Hematol Oncol 2008 May;30(5):387-390.
4. Tanaka M, Fukushima N, Itamura H, Urata C, Yokoo M, Ide M, et al. Gangrenous cheilitis associated with all-trans retinoic acid therapy for acute promyelocytic leukemia. Int J Hematol 2010 Jan;91(1):132-135.
5. Frankel SR, Eardley A, Lauwers G, Weiss M, Warrell RP Jr. The "retinoic acid syndrome" in acute promyelocytic leukemia. Ann Intern Med 1992 Aug;117(4):292-296.
6. Ahmed Z, Shaikh MA, Raval A, Mehta JB, Byrd RP Jr, Roy TM. All-trans retinoic acid syndrome: another cause of drug-induced respiratory failure. South Med J 2007 Sep;100(9):899-902.
7. Tazi I, Rachid M, Quessar A, Benchekroun S. Scrotal ulceration following all-trans retinoic Acid therapy for acute promyelocytic leukemia. Indian J Dermatol 2011 Sep-Oct;56(5):561-563.
8. Mori A, Tamura S, Katsuno T, Nishimura Y, Itoh T, Saheki K, et al. Scrotal ulcer occurring in patients with acute promyelocytic leukemia during treatment with all-trans retinoic acid. Oncol Rep 1999 Jan-Feb;6(1):55-58.
9. Goto H, Tsurumi H, Kasahara S, Hara T, Yamada T, Sawada M, et al. [Acute promyelocytic leukemia accompanied by scrotal Fournier’s gangrene during ATRA treatment and relapsed as external ear tumor]. Rinsho Ketsueki 1998 Dec;39(12):1169-1174.
10. Sun GL. [Treatment of acute promyelocytic leukemia (APL) with all-trans retinoic acid (ATRA): a report of five-year experience]. Zhonghua Zhong Liu Za Zhi 1993 Mar;15(2):125-129.
11. Shimizu D, Nomura K, Matsuyama R, Matsumoto Y, Ueda K, Masuda K, et al. Scrotal Ulcers Arising during Treatment with All-trans Retinoic Acid for Acute Promyelocytic Leukemia. Intern Med 2005 May;44(5):480-483.
12. Tajima K, Sagae M, Yahagi A, Akiba J, Suzuki K, Hayashi T, et al. [Scrotum exfoliative dermatitis with ulcers associated with treatment of acute promyelocytic leukemia with all-trans retinoic acid]. Rinsho Ketsueki 1998 Jan;39(1):48-52.
13. Lévy V, Jaffarbey J, Aouad K, Zittoun R. Fournier’s gangrene during induction treatment of acute promyelocytic leukemia, a case report. Ann Hematol 1998 Feb;76(2):91-92.
14. Fukuno K, Tsurumi H, Goto H, Oyama M, Tanabashi S, Moriwaki H. Genital ulcers during treatment with ALL-trans retinoic acid for acute promyelocytic leukemia. Leuk Lymphoma 2003 Nov;44(11):2009-2013.
15. Degos L, Dombret H, Chomienne C, Daniel MT, Micléa JM, Chastang C, et al. All-trans-retinoic acid as a differentiating agent in the treatment of acute promyelocytic leukemia. Blood 1995 May;85(10):2643-2653.
16. Koga H, Fujita I, Miyazaki S. Effects of all-trans-retinoic acid on superoxide generation in intact neutrophils and a cell-free system. Br J Haematol 1997 May;97(2):300-305.
17. Arun B, Berberian B, Azumi N, Frankel SR, Luksenburg H, Freter C. Sweet’s syndrome during treatment with all-trans retinoic acid in a patient with acute promyelocytic leukemia. Leuk Lymphoma 1998 Nov;31(5-6):613-615.
|