|
Abstract
Objective: Nuclear factor kappa B (NF-κB) pathway and oxidative stress participate in endothelial dysfunction, which is one of the causes of pre-eclampsia. Among the human antioxidant mechanisms, there are the enzymes catalase (CAT), glutathione peroxidase (GPx) and superoxide dismutase (SOD). Our aim was to measure NF-κB, its inhibitor (IKK) and oxidative stress in placenta and umbilical cord of pregnant women submitted to a supervised nutritional program.
Methods: Two groups were conformed: A) 14 pregnant women with individualized nutritional counseling, and B) 12 pregnant women without nutritional guidance. NF-κB and IKK were assessed by real time PCR (RT-PCR). Enzymatic activity of CAT, GPx, lipoperoxidation (LPO) and SOD were also evaluated.
Results: Pregnant women that followed a supervised nutritional program had lower levels of systolic (p=0.03) and diastolic pressure (p=0.043) although they were heavier than the control group (p=0.048). Among all the women, the Spearman correlation was positive between weight gain and placental NF-κB expression (1, p≤0.01). In the placenta, women with nutritional advice had lower enzymatic activity of GPx (p≤0.038) and showed a tendency of IKK to be higher than in women without a nutritional supervised program.
Conclusion: A supervised nutritional program in pregnancy offers a proven option to control weight gain, hypertension, NF-κB/IKK complex expression and oxidative stress reactions in the placenta.
Keywords: Diet; NF-κB; Oxidative stress; Pre-eclampsia; Pregnancy.
Introduction
Obesity during pregnancy is associated with exaggerated metabolic adaptation, endothelial dysfunction and an increased risk of adverse pregnancy outcomes, including pre-eclampsia,1 hypertension and gestational diabetes mellitus (GDM).2 Pre-eclampsia is a major cause of maternal and fetal morbidity and mortality that affects approximately 3% to 7% of all pregnant women.3
Nuclear factor-kappa B (NF-κB), a heterodimer located in the cytoplasm, is bound by its inhibitor κB (IκB), which prevents NF-κB from entering the nucleus.4 The activation of a systemic inflammatory process demonstrated in plasma from pre-eclamptic women has been attributed to NF-κB, by promoting the synthesis of cytokines, interleukin (IL)-6, IL-8, monocyte chemotactic protein-1 (MCP-1) and expression of intercellular adhesion molecule (ICAM-1).5
There is current evidence indicating that NF-κB is activated by reactive oxygen species (ROS), [superoxide anions (•O2-), hydrogen peroxide (H2O2) and hydroxyl radicals (•OH)].6 Besides, ROS interact with proteins, nucleic acids and lipids, as well as in a process called lipid peroxidation (LPO), that can cause severe cell and tissue damage.7,8
To maintain balance of the oxidative state, the human body possesses intracellular antioxidant enzyme systems, including catalase (CAT), five different forms of glutathione peroxidase (GPx) and three isoforms of superoxide dismutase (SOD).9 Under physiological conditions, these defense mechanisms maintain a low concentrations of ROS within the cell and regulate ROS activity. Thus, a balance between ROS production and antioxidant defenses determines the degree of oxidative stress.10
The role of maternal diet in the etiology of pre-eclampsia has received increasing attention.11 Based on the epidemiology of pre-eclampsia, which is more common among poor women, the findings had previously suggested that nutrients may be involved in the disorder, unfortunately, the nutritional data obtained from women with the syndrome has been poorly defined.12
A current theory holds that an imbalance between maternal pro- and anti-oxidants, is a component of pre-eclampsia.13 It still remains unknown whether such an imbalance occurs prior to clinical recognition of the syndrome or whether it is related to diet; however, maternal diet is a known, underlying environmental factor that contributes to free radical generation.14
The objective of this paper was to measure NF-κB, its inhibitor subunit beta (IKKB), and oxidative stress in the placenta and umbilical cord of pregnant women submitted to a supervised nutritional program, in order to provide a possible explanation of the benefits of limiting gestational weight gain.
Methods
This clinical, prospective and comparative study was carried out in Toluca, Mexico, during March 2009 to December 2010. Pregnant women aged 18-40 years in the first trimester of pregnancy, attended the Maternal-Perinatal Hospital "Mónica Pretelini" (HMPMP), Toluca, Mexico, were considered for the study. Subjects with multiple pregnancies or prior history of chronic-metabolic diseases including GDM or pre-pregnancy hypertension were excluded. Women with incomplete medical records or those who did not deliver at the hospital or attended less than two nutritional visits during the pregnancy, were considered lost for followup of the study.
Two study groups were formed: A) women who were offered an individualized diet, and B) women attending the obstetrics unit who did not follow a supervised diet. Initially, we assigned 20 pregnant women to each group (actually, 13 were enough to recognize a statistically significant difference ≥2 relative units of NF-κB, with α risk of 0.05 and a β risk of 0.2 in a two-sided test, with a standard deviation (SD) of 1.7 and a drop-out rate of 10%),15 from whom 14 completed the nutritional supervision and 12 constituted the control group (one patient did not come back, nine deliveries were attended in other hospitals and four were discarded because samples were not well preserved).
All women who sought medical attention at the high-risk pregnancy clinic of the HMPMP were offered the opportunity to be included in the study. After the acceptance of one mother to be nutritionally supervised, we captured one control patient in the Red Code Service with the most proximal age to the case. If this woman was lost in the follow-up period, another woman was we captured in the previous service without age criteria due to time limitations (nine months of pregnancy).
Anthropometric measures were assessed in the high-risk pregnancy clinic by HMPMP nutritionist. Body weight was measured in an overnight fasting status without shoes in a minimal clothing state by the use of a digital scale (Seca, Hamburg, Germany) to the nearest 0.1 kg. Height was measured using a non-stretched tape measure (Seca, Hamburg, Germany) to the nearest 0.1 cm. Pre-pregnancy BMI was calculated as weight in kg divided by height in meters squared based on the prenatal chart or self-reported weight in women with no prenatal chart, since previous studies have ratified its suitability.16
Blood pressure was recorded at each visit using a standard sphygmomanometer (Riester Big Ben® Square, Germany) and appropriately sized cuff, that was changed to an electronic monitor (Mercury, Mennem Medical, USA) if a case was confirmed as preeclampsia.
Fasting blood samples (10 mL) were taken at the HMPMP laboratory in the early morning after an overnight fast. Serum samples were analyzed for serum albumin, calcium, cholesterol, creatinine, magnesium, triglycerides and uric acid using appropriate kits (Dimension Rx L Max, Dade Behring, USA). Serum hemoglobin was measured using available kits (Advia 120, Bayer Health, USA). Fasting plasma glucose (FPG) was assayed with available kits (Dimension Rx L Max, Dade Behring, USA).
Dietetic treatment was calculated according to height, weeks of gestation and weight, considering an energy intake of 30 kcal/kg of expected weight, distributing the resulting energy according to the percentage of macronutrients’ adequation (55-65% carbohydrates, 10-20% fat and the remainder as proteins).17 On each nutritional visit, 24 hour dietary recall was done and analyzed using NutriKcal®VO software in order to evaluate the Healthy Eating Index (HEI), which is commonly employed to assess pregnant women’s dietary adequacy (on a scale from 0-100), through the consumption of 12 components of food groups described previously.18,19 An improvement in diet was considered when the initial HEI score improved more than one point during the next nutritional assessment.
Pre-eclampsia was diagnosed and classified according to the criteria specified by the technical bulletin of the American College of Obstetricians and Gynecologists (ACOG) and the National High Blood Pressure Education Program (NHBPEP) Working Group Report on High Blood Pressure in Pregnancy.20
During pregnancy resolution, a sample of placenta and umbilical cord in heparinized vials were kept at -80°C (Forma -86ºC ULT Freezer, Thermo Electron Corporation, USA) until use. One sample of each tissue from all mothers was weighed (Ohaus Trip Balance), prepared in buffer under sterile conditions (Forma Laminar Airflow Workstation, Thermo Scientific), heated (J18 Bain Univ, Jouan) and centrifuged (IEC CL40R Centrifuge, Thermo Scientific).
mRNA was isolated using the Magna Pure LC RNA Isolation Kit III and retro-transcribed with the Transcriptor High Fidelity cDNA Synthesis Kit (Roche) to obtain cDNA. Samples were then quantified using a nano spectrophotometer set (Nano Photometer, Implen) at two wavelengths (260 nm and 280 nm), with an acceptable degree of purity between 1.8 and 2 for real time reverse transcriptase polymerase chain reaction (RT-PCR) performed in a Roche LightCycler 2.0 using the SYBR® Advantage® qPCR Premix Kit following the manufacturer’s instructions. The steps were: denaturation, alignment and elongation. Primer sequences (determined with Primer 3 software) were as follows: NF-κB (NCBI: NM_003998.3), fw: 5’-cctggatgactcttgggaaa-3’, rv: 5’-tcagccagctgtttcatgtc-3’ and IKKB (NCBI: NM_001556.1), fw: 5’-gctgcaactgatgctgatgt-3’, rv: 5’-tgtcacagggtaggtgtgga-3’. The relative expression of each gene, compared to expression of the beta-2 microglobulin (B2M) gene (NCBI: NM_004048.2) (control) using fw: 5’-ttcatccstccgacattgaa-3’ and rv: 5’-cctccatgatgctgcttaca-3’ was calculated through the method 2ΔΔCT.
RT-PCR products were performed at the Laboratory of Molecular Biology, Medical Research Center (CICMED), Autonomous University of the State of Mexico (UAEMex) and sequenced and read with Chromas software v. 2.33 (Instituto de Neurobiología, National Autonomous University of Mexico, UNAM), to verify the correct amplification of the primers.
Tissue (1 g) was added into 4 mL of Phosphate Buffer Solution (PBS) (pH 7.4) and homogenized (Tissue Tearor™, Biospec Products, Inc.). The supernatant was then centrifuged at 2000 rpm for 5 min and then at 9000 rpm for 15 min. Next, it was sonicated for 30 min and then submitted to a final centrifugation step at 13,500 rpm for 30 min. The entire process was conducted at -4ºC and the supernatant was removed and used for different determinations.
CAT activity (μmol H2O2/mg protein) was determined according to the technique described by Radi et al.21 GPx activity (nmol NADPH/mg protein) was quantified by the Paglia and Valentine method.22 LPO (nmol MAD/mg protein) was determined using the Büege and Aust method.23 SOD activity (mmol/mg protein) was analyzed according to the Misra and Fridovich modified method.24
Protein concentration was spectrophotometrically determined using the Bradford protein assay.25 To 25 μL of supernatant, 75 μL deionized water and 2.5 mL of Bradford’s reagent (100 mg Coomassie Blue dye, 50 mL 95% ethanol and 100 mL H3PO4, in 1 L deionized water) were added. Tubes were shaken and allowed to rest for 5 min prior to reading the absorbance at 595 nm and interpolation using a bovine serum albumin standard curve. Enzymatic and protein quantification were performed at the Faculty of Chemistry, UAEMex.
Written consent was obtained from all subjects. This study was approved by the Bioethics and Research Committee of the HMPMP, date code 05/06/09.
Statistical analysis of the data was carried out using SPSS version 17. Continuous variables were expressed as mean ± SD. As there was no normal distribution, the groups were compared using the Mann-Whitney U-test. The degree of association between the biomarkers of oxidative stress with gene expression was evaluated using Spearman’s correlation; p<0.05 was considered statistically significant.
Results
Table 1 lists the results of maternal characteristics of the study population. In women receiving nutritional advice, from whom nine (64.2%) had improvement in the quality of diet, statistically significant differences were seen in weeks of gestation (weeks of pregnancy from the start of the last normal menstrual period) (longer, p≤0.01), pre-pregnancy weight and pre-pregnancy BMI (p≤0.05). At the end of their gestational period, women in the group without nutritional advice had higher systolic, diastolic and median hypertension levels (p≤0.05). Six women (42.8%) from group A (with individualized diets) had pre-gestational obesity (pre-pregnancy BMI 330 kg/m2) and two women (14.2%) from this group developed pre-eclampsia. Unsurprisingly, these patients had the second and third highest weight gain (12 and 11 kg), which were 2-3 kg above the median (9 kg). Eight women (57.1%) from this group were pre-gestationally overweight (25 < pre-pregnancy BMI ≤30) and none of them had a normal pre-pregnancy BMI (<25).
From group B (no diet supervision), only one woman (8.3%) had pre-gestational obesity, and three (25%) were overweight. A total of six women from this group developed pre-eclampsia, which included the individuals with the first and third highest weight gain.
In the placenta, IKK showed a tendency to be higher in pregnancies followed-up with a supervised diet (Fig. 1). Notwithstanding, this tendency did not reach a statistically significant difference (p=0.32). NF-κB showed no significant differences between the groups. In the umbilical cord, NF-κB was poorly detected, this may be attributed to its rich content in Wharton's jelly and IKK did not present any significant differences.
Table 1: Clinical and anthropometrical characteristics.1
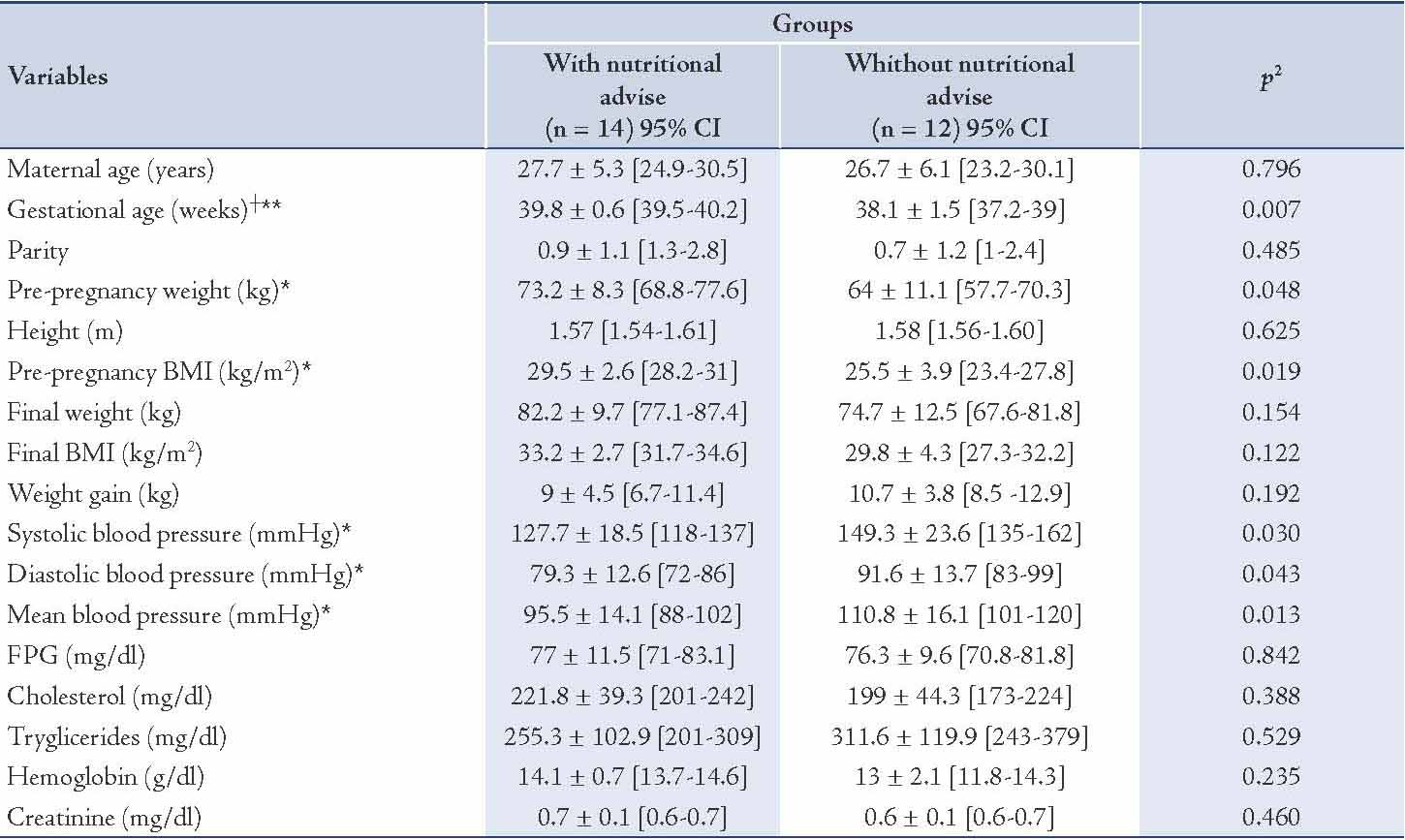
Table 2: Enzymatic activity in the placenta and umbilical cord
Figure 1: In plaventa, IKK showed a tendancy to be higher in pregnancies.
There were no differences in CAT, LPO and SOD activity. Furthermore, there was no difference with the oxidant/antioxidant status formula: SOD/(CAT+GPx). Only a statistically significant difference in GPx enzymatic activity in placenta was observed (p≤0.05), with higher values in the group without nutritional advice. (Table 2)
Among all women, the Spearman correlation was positive between pre-pregnancy weight and final weight (0.934, p≤0.001) and between pre-pregnancy BMI and final weight (0.663, p=0.001); with placental values this correlation was positive between pre-pregnancy BMI and SOD (0.341, p=0.018), between weight gain and NF-κB (1, p≤0.001), between CAT and GPx (0.634,p≤0.001) and negative between LPO and IKK (-0.603, p=0.013).
Significant positive intra-group correlations in those women that completed the nutritional supervision were found between pre-pregnancy weight and BMI (0.739, p=0.003), final weight (0.884, p≤0.001), final BMI (0.657, p=0.011) and history of cesareans (0.582, p=0.037), between BMI and history of pregnancies (0.633, p=0.020) and cesareans (0.838, p=0.001) and final BMI (0.825, p≤0.001), between umbilical cord determinations of LPO and GPx (0.955), H2O2 (0.925) and SOD (0.999) all three with p≤0.001. On the other hand, negative correlations were found between placental IKK and hemoglobin (-0.845, p=0.034).
Finally, in the group without nutritional advice, significant correlations were found between age and final BMI (0.645, p=0.044), between weeks of pregnancy and cholesterol (0.960, p=0.002), between pre-pregnancy weight and BMI (0.949, p=0.003), final weight (0.954, p≤0.001) and final BMI (0.911, p≤0.001), between BMI and final weight (0.882, p=0.001) and final BMI (0.943, p≤0.001), between triglycerides and GPx (in placenta 0.948, p=0.014, in umbilical cord 0.980, p=0.003) and H2O2 (in placenta 0.941, p=0.017, in umbilical cord 0.959, p=0.01), between placental GPx and umbilical cord GPx (0.953) and H2O2 (0.940) both with p≤0.001.
As a dependent variable, placental H2O2 reached statistical significant differences with weight gain and mean blood pressure (p=0.001). NADPH showed the same behavior with weight gain (p=0.001). Also, umbilical cord H2O2 showed statistical significant difference with weight gain (p=0.001). Data for NF-κB was suggestive but missed statistical significance (p=0.053).
Discussion
It is recognised that the action of maternal body composition and diet can influence outcomes of pregnancy and early childhood.26 In fact, obesity upregulates, placenta proinflammatory pathways and oxidative stress.27 The majority of these effects are mediated through the p38 mitogen-activated protein kinases (p38 MAPK) and NF-κB pathways.4,28 But at present, most of the papers have published results from newborns or umbilical cord cells,29 underestimating the contribution made by maternal BMI and diet to feto-placental oxidative stress.30
The limitation in weight gain has been proven to protect against obesity related complication of pregnancy such as all types of hypertension, GDM and even fetal loses. In our study, it is interesting to note that although the group with nutritional advice was heavier at the beginning of pregnancy; in the end, this group had lower weight gain, systolic and diastolic blood pressures compared to the group without nutritional advice. The improved metabolic control could explain in part a longer gestational period in the group with nutritional advice.
Measuring the adherence to nutritional therapy is essential to evaluate whether outcomes are related to any given recommendations. In the current study, we verified diet quality improvement in nine of 14 women submitted to the supervised nutritional program (64.2%). Several indexes have been proposed to assess diet quality in pregnancy and its effects on health outcomes.18,31,32 However, the use of such indexes to evaluate oxidative stress in the placenta is rare. This work suggests that NutriKcal®VO software is useful in pregnacy in order to evaluate the HEI index.
Given the large number of publications related to the NF-kB/IKK complex,33 and the increasing attention of its expression in the placenta,34-36 it is surprising that an explanation of how this complex is modulated by the mother’s diet is still unclear. This is most likely because the extremely difficult process to standardize and supervise food intake during pregnancy. In our study, the group with nutritional guidance showed a tendency to have higher levels of IKK, suggesting a beneficial effect on the inflammatory pathways by preventing NF-kB signaling into the nucleus. This finding represents a necessary, but not sufficient role of IKK in preventing oxidant stress effects,37 as more recently, it has been shown that the long-standing model of cytoplasmic sequestration by IkB proteins is only partially true.4
Despite a lack of statistical difference in NF-κB between the two groups, there was a clear correlation with weight gain during pregnancy. Further work is required to define in detail the modulation of NF-κB activity with different types of diets during pregnancy.
It has already been published that SOD is significantly decreased, while LPO (MDA) is increased in pre-eclamptic cases compared to normal pregnant women.38 Furthermore, GPx activity in pre-eclamptic placenta has been shown to be both downregulated39 and upregulated.40 Notwithstanding these conflicting data, it is clear that improving maternal diet during pregnancy is beneficial for the placental and fetal antioxidant capacities.41
Taking into account the above studies, we decided to evaluate the oxidative stress state in the placenta and umbilical cord following/not following a gestational supervised diet. Surprisingly, neither CAT nor SOD was up-regulated in women without nutritional advice. Rather, the response was quite similar in both groups. The only difference found in this sense was with GPx, which was in concordance with those results reported by other authors in plasma,38 erythrocytes,42 and placenta.40 The oxidant/antioxidant status formula was almost statistically significant in umbilical cord (p=0.52), which shows how sensitive this tissue might be to ROS and inflammation. Interestingly, the intra-group correlations suggest that the diet intervention could contribute to diminish oxidative stress by limiting the triglycerides increase.
Of note, multiple regression analysis shown that weight gain predicts oxidative stress, considering H2O2 levels activity. Adding this to the positive correlations between pre-pregnancy weight and final weight and BMI, supports the importance of controlling weight in benefit of the patients.
Within the limitations of this study, it has been showed that it is possible to get improvement in the quality of diet during pregnancy if a nutritional supervision is offered. Taken together, our data indicate that nutrition counseling has a positive impact on the health of women with high-risk pregnancies and limiting weight gain as reported by others,43 besides preventing clinical complications due to nutritional deficiences.44 Yet, another hypothesis is the putative beneficial effect of a quantitative and qualitative supervised mother’s diet, by keeping NF-κB joined to its inhibitor, thus preventing the effects of oxidative stress in the placenta.
This study does have its limitations. First, the HMPMP is a high-risk pregnancy reference hospital and the conclusions may not be applicable to other populations. Second, despite our best efforts to establish a good coordination with the patients, nutritionists and medical staff, the number of study patients was low because mothers may have sought medical attention at other hospitals.
Conclusion
We, therefore suggest that additional studies on the nutritional guidance of high-risk pregnancies are needed to investigate the oxidative status modulation by different individualized interventions and its relation to pregnancy complications.
Acknowledgements
Authors thank the next people for taking samples: Luis Fernando Arias Lana, Adriana Casas Navarro, Emilio Martínez Fernández, Cristina Meléndez Zambrano, Hilmara Ramírez López. This work was supported by the National Council of Science and Technology (CONACYT)-Mexico (grant number: 95558) and Secretariat of Public Education (SEP) (PROMEP, grant number: FE02/2010, project: 734202), Mexico.
Clinical trials ID: NCT01667939.
References
1. Jarvie E, Hauguel-de-Mouzon S, Nelson SM, Sattar N, Catalano PM, Freeman DJ. Lipotoxicity in obese pregnancy and its potential role in adverse pregnancy outcome and obesity in the offspring. Clin Sci (Lond) 2010 Aug;119(3):123-129.
2. Ethier-Chiasson M, Forest JC, Giguère Y, Masse A, Marseille-Tremblay C, Lévy E, et al. Modulation of placental protein expression of OLR1: implication in pregnancy-related disorders or pathologies. Reproduction 2008 Oct;136(4):491-502.
3. Duarte M, Espinosa L, Díaz M, Sánchez R, Lee Eng C, Díaz C. Preeclampsia y desequilibrio del estado óxido-reduccción. Papel de los antioxidantes. Med Int Mex 2008;24(6):407-413.
4. Tergaonkar V, Correa RG, Ikawa M, Verma IM. Distinct roles of IkappaB proteins in regulating constitutive NF-kappaB activity. Nat Cell Biol 2005 Sep;7(9):921-923.
5. Cindrova-Davies T, Spasic-Boskovic O, Jauniaux E, Charnock-Jones DS, Burton GJ. Nuclear factor-kappa B, p38, and stress-activated protein kinase mitogen-activated protein kinase signaling pathways regulate proinflammatory cytokines and apoptosis in human placental explants in response to oxidative stress: effects of antioxidant vitamins. Am J Pathol 2007 May;170(5):1511-1520.
6. Morgan MJ, Liu ZG. Crosstalk of reactive oxygen species and NF-κB signaling. Cell Res 2011 Jan;21(1):103-115.
7. Rajasingam D, Seed PT, Briley AL, Shennan AH, Poston L. A prospective study of pregnancy outcome and biomarkers of oxidative stress in nulliparous obese women. Am J Obstet Gynecol 2009 Apr;200(4):e1-e9.
8. Rizzo AM, Berselli P, Zava S, Montorfano G, Negroni M, Corsetto P, et al. Endogenous antioxidants and radical scavengers. Adv Exp Med Biol 2010;698:52-67.
9. Jomova K, Valko M. Advances in metal-induced oxidative stress and human disease. Toxicology 2011 May;283(2-3):65-87.
10. Avery SV. Molecular targets of oxidative stress. Biochem J 2011 Mar;434(2):201-210.
11. Xu H, Shatenstein B, Luo ZC, Wei S, Fraser W. Role of nutrition in the risk of preeclampsia. Nutr Rev 2009 Nov;67(11):639-657.
12. Roberts JM, Balk JL, Bodnar LM, Belizán JM, Bergel E, Martinez A. Nutrient involvement in preeclampsia. J Nutr 2003 May;133(5)(Suppl 2):1684S-1692S.
13. Poston L. Endothelial dysfunction in pre-eclampsia. Pharmacol Rep 2006;58(Suppl):69-74.
14. Scholl TO, Leskiw M, Chen X, Sims M, Stein TP. Oxidative stress, diet, and the etiology of preeclampsia. Am J Clin Nutr 2005 Jun;81(6):1390-1396.
15. Granmo software. From: http://www.imim.cat/ofertadeserveis/software-public/granmo/. Accessed: Aug 2012.
16. Casanueva E, Flores-Quijano ME, Roselló-Soberón ME, De-Regil LM, Sámano R. Gestational weight gain as predicted by pregestational body mass index and gestational age in Mexican women. Food Nutr Bull 2008 Dec;29(4):334-339.
17. Casanueva E, Flores Quijano ME. Nutrición de la Mujer Adulta. In: Casanueva E, Kaufer-Horwitz M, Pérez-Lizaur AB, rroyo P eds. Nutriología Médica. Mexico: Panamericana. 2008: 174-210.
18. Pick ME, Edwards M, Moreau D, Ryan EA. Assessment of diet quality in pregnant women using the Healthy Eating Index. J Am Diet Assoc 2005 Feb;105(2):240-246.
19. Tsigga M, Filis V, Hatzopoulou K, Kotzamanidis C, Grammatikopoulou MG. Healthy Eating Index during pregnancy according to pre-gravid and gravid weight status. Public Health Nutr 2011 Feb;14(2):290-296.
20. Report of the National High Blood Pressure Education Program Working Group on High Blood Pressure in Pregnancy. Report of the National High Blood Pressure Education Program Working Group on High Blood Pressure in Pregnancy. Am J Obstet Gynecol 2000 Jul;183(1):S1-S22.
21. Radi R, Turrens JF, Chang LY, Bush KM, Crapo JD, Freeman BA. Detection of catalase in rat heart mitochondria. J Biol Chem 1991 Nov;266(32):22028-22034.
22. Paglia DE, Valentine WN. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 1967 Jul;70(1):158-169.
23. Buege JA, Aust SD. Microsomal lipid peroxidation. Methods Enzymol 1978;52:302-310.
24. Misra HP, Fridovich I. The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem 1972 May;247(10):3170-3175.
25. Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976 May;72:248-254.
26. Perichart-Perera O, Balas-Nakash M, Parra-Covarrubias A, Rodríguez-Cano A, Ramírez-Torres A, Ortega-González C, et al. A medical nutrition therapy program improves perinatal outcomes in Mexican pregnant women with gestational diabetes and type 2 diabetes mellitus. Diabetes Educ 2009 Nov-Dec;35(6):1004-1013.
27. Roberts VH, Smith J, McLea SA, Heizer AB, Richardson JL, Myatt L. Effect of increasing maternal body mass index on oxidative and nitrative stress in the human placenta. Placenta 2009 Feb;30(2):169-175.
28. Baker RG, Hayden MS, Ghosh SN. NF-κB, inflammation, and metabolic disease. Cell Metab 2011 Jan;13(1):11-22.
29. Sarikabadayi YU, Aydemir O, Aydemir C, Uras N, Oguz SS, Erdeve O, et al. Umbilical cord oxidative stress in infants of diabetic mothers and its relation to maternal hyperglycemia. J Pediatr Endocrinol Metab 2011;24(9-10):671-674.
30. Thornburg KL, O’Tierney PF, Louey S. Review: The placenta is a programming agent for cardiovascular disease. Placenta 2010 Mar;31(Suppl):S54-S59.
31. Bodnar LM, Siega-Riz AM. A Diet Quality Index for Pregnancy detects variation in diet and differences by sociodemographic factors. Public Health Nutr 2002 Dec;5(6):801-809.
32. Rodríguez-Bernal CL, Rebagliato M, Iñiguez C, Vioque J, Navarrete-Muñoz EM, Murcia M, et al. Diet quality in early pregnancy and its effects on fetal growth outcomes: the Infancia y Medio Ambiente (Childhood and Environment) Mother and Child Cohort Study in Spain. Am J Clin Nutr 2010 Jun;91(6):1659-1666.
33. Hayden MS, Ghosh SN. NF-κB, the first quarter-century: remarkable progress and outstanding questions. Genes Dev 2012 Feb;26(3):203-234.
34. Zhu MJ, Du M, Nathanielsz PW, Ford SP. Maternal obesity up-regulates inflammatory signaling pathways and enhances cytokine expression in the mid-gestation sheep placenta. Placenta 2010 May;31(5):387-391.
35. Li M, Wu ZM, Yang H, Huang SJ. NFκB and JNK/MAPK activation mediates the production of major macrophage- or dendritic cell-recruiting chemokine in human first trimester decidual cells in response to proinflammatory stimuli. J Clin Endocrinol Metab 2011 Aug;96(8):2502-2511.
36. Lappas M, Yee K, Permezel M, Rice GE. Lipopolysaccharide and TNF-alpha activate the nuclear factor kappa B pathway in the human placental JEG-3 cells. Placenta 2006 Jun-Jul;27(6-7):568-575.
37. Wright CJ, Agboke F, Muthu M, Michaelis KA, Mundy MA, La P, et al. Nuclear factor-κB (NF-κB) inhibitory protein IκBβ determines apoptotic cell death following exposure to oxidative stress. J Biol Chem 2012 Feb;287(9):6230-6239.
38. Chamy VM, Lepe J, Catalán A, Retamal D, Escobar JA, Madrid EM. Oxidative stress is closely related to clinical severity of pre-eclampsia. Biol Res 2006;39(2):229-236.
39. Vanderlelie J, Venardos K, Clifton VL, Gude NM, Clarke FM, Perkins AV. Increased biological oxidation and reduced anti-oxidant enzyme activity in pre-eclamptic placentae. Placenta 2005 Jan;26(1):53-58.
40. Knapen MF, Peters WH, Mulder TP, Merkus HM, Jansen JB, Steegers EA. Glutathione and glutathione-related enzymes in decidua and placenta of controls and women with pre-eclampsia. Placenta 1999 Sep;20(7):541-546.
41. Lin Y, Han XF, Fang ZF, Che LQ, Nelson J, Yan TH, et al. Beneficial effects of dietary fibre supplementation of a high-fat diet on fetal development in rats. Br J Nutr 2011 Aug;106(4):510-518.
42. Krishna Mohan S, Venkataramana G. Status of lipid peroxidation, glutathione, ascorbic acid, vitamin E and antioxidant enzymes in patients with pregnancy–induced hypertension. Indian J Physiol Pharmacol 2007 Jul-Sep;51(3):284-288.
43. Asbee SM, Jenkins TR, Butler JR, White J, Elliot M, Rutledge A. Preventing excessive weight gain during pregnancy through dietary and lifestyle counseling: a randomized controlled trial. Obstet Gynecol 2009 Feb;113(2 Pt 1):305-312.
44. Samimi M, Asemi Z, Taghizadeh M, Azarbad Z, Rahimi-Foroushani A, Sarahroodi S. Concentrations of Serum Zinc, Hemoglobin and Ferritin among Pregnant Women and their Effects on Birth Outcomes in Kashan, Iran. Oman Med J 2012 Jan;27(1):40-45.
|