Type I diabetes mellitus (T1DM) is one of the most common chronic diseases affecting around 500,000 children globally.1 Despite improved insulin delivery systems, greater availability of various insulin types, and the development of new treatment protocols, only 30% of children with T1DM achieve glycated hemoglobin (HbA1c) values below eight.2
The Diabetes Control and Complication Trial (DCCT) clearly demonstrated that tight metabolic control is a crucial element in the prevention of micro- and macrovascular complications.2 Furthermore, optimizing glycemic control in children with T1DM is a key factor for attaining normal growth and pubertal development. The use of intensive insulin treatment in the form of multiple daily injections (MDI) has been the standard treatment method in most clinical practice settings worldwide. This method has been applied from the onset of disease in both adolescents and young diabetic patients.3 As noted in the DCCT trials,2 the main drawbacks of intensive insulin therapy were weight gain and an increase in severe hypoglycemia events.
Although it has been available since the late 1970’s, continuous subcutaneous insulin infusion (CSII) has only been widely used in pediatric patients since 2000.4 More recently, there has been a significant increase in CSII use in children and adolescents4 with variable reported effects on glycemic control.5-8 CSII has been shown to achieve a significantly lower risk of severe hypoglycemia with no increase in the risk of diabetic ketoacidosis.8-12
The incidence of T1DM in children and adolescents in Kuwait has been rising dramatically. In 2011, the incidence calculated from the national diabetes registry13 was 38.9 per 100,000. Every child diagnosed with T1DM is treated and followed-up in the pediatric diabetes clinics in one of six government hospitals.
In 2007, the Ministry of Health approved full financial coverage for all Kuwaiti nationals children and adolescents with T1DM and issued guidelines for its use in this population group. The insulin pump committee, established by the Ministry, set certain criteria for pump eligibility. Patients had to: a) have T1DM, b) demonstrate the ability to self-monitor blood glucose level at least four times per day, c) demonstrate compliance with dietary plans and insulin regimens, and d) successfully attend and pass a carbohydrate-counting course. Glycemic control assessed by HbA1c level was not a criterion for eligibility.
Non-nationals were able to obtain a pump through The Patient Helping Fund Society or other charity agencies in the country.
The aim of our study was to assess the impact of CSII use in children and adolescents on glycemic control, insulin dose requirement, and body mass index (BMI) compared to children using a MDI regimen.
Methods
Between July 2007 and December 2012, 512 patients’ aged 18 years old or above with T1DM underwent insulin pump initiation in one of the six governorate hospitals or the Dasman Diabetes Center in Kuwait. Others remained on MDIs. Data on 326 subjects were retrospectively included in the study analysis.
The following inclusion criteria were applied: availability of pre-pump clinical and biochemical data (insulin requirements, HbA1c level, weight, and height) as well as complete follow-up data; CSII started based on the criteria of the Ministry of Health pump committee and followed-up in a government hospital (i.e. not the private health sector); and if the patient had been on CSII for at least six months at the time of the study (to minimize the effect of the honeymoon period), and were not using long-acting insulin with CSII.
T1DM was diagnosed based on International Society of Pediatric and Adolescent Diabetes/International Diabetes Federation (ISPAD/IDF) guidelines.14 Each patient in the CSII group was matched for age, sex and diabetes duration with a patient on MDI (n=326).
The MDI protocol consisted of intensive insulin therapy (three to four injections per day) of long and short-acting analogs (glargine and aspart) and home blood glucose measurement (three to four times per day). Patients on MDI were trained in advanced carbohydrate counting using grams, as well as food label reading, similar to the pump group. Good glycemic control was neither a criterion for pump therapy nor study inclusion. All centers prescribed the Paradigm 722 and Veo Medtronic MiniMed Solution pump with CareLinkTM (Medtronic MiniMed, Inc., California, US). Only five (of the 522) patients were using the continuous glucose monitoring system and they were not included in the analysis. All pumps except one used ultra-short-acting insulin, aspart.
Data were collected from patients’ medical charts. Patients were seen routinely every four to six weeks during MDI and after the pump therapy stabilization period. BMI, insulin dose, HbA1c, and severe hypoglycemia frequency (self-reports) and diabetic ketoacidosis (DKA) were evaluated at baseline and every three months for up to five years. Skin infections, allergy, bleeding, and bruising were recorded.
At each clinic visit, the patient’s weight and height were measured, and BMI was calculated using the official formula. BMI standard deviation scores (z-scores) were used for analysis. HbA1c values were measured for all patients at the clinic visits every three months.
HbA1c values were aligned with the DCCT (normal range 4.4–6.3%, mean 5.4%, and interassay SD 0.15%) using the Tosoh Analyzer (Tosoh Bioscience, Inc., San Francisco, USA). Insulin (units per kg per day) were calculated at each visit for all patients. A severe hypoglycemic episode was defined as a decreased level of consciousness and/or seizure requiring assistance from others, glucagon use, and hospital admission. Diabetic ketoacidosis (DKA)was defined as a pH greater than 7.3, ketonuria or ketonemia, and bicarbonate levels greater than 15mmol/L.14
CSII initiation was preceded by a training program for patients and their caregivers on pump technology. Certified pump trainers conducted the training in a hospital outpatient setting. In some places the training was done by educators from the company supplying the pumps. The program covered principles of pump operation, quick set insertion and care, carbohydrate counting, and insulin bolusing. Patients were advised to perform 6–8 blood glucose tests: before and after meals, at midnight, and at 3 a.m. All patients and their caregivers in both groups received intensive training by a dietician on carbohydrate counting and reading food labels.
Initially, the total dose for CSII was calculated as 75% of the total daily insulin dose (TTD) on MDI. The initial setting was 50% of the dose as basal requirements for 24 hours and 50% as boluses. Subsequently, insulin doses were adjusted based on the subjects’ blood glucose patterns and daily activity routines. Patients were instructed to change the infusion sets every three days and when skin infections or irritation was noticed. Catheter occlusion was considered when high blood glucose persisted and did not respond to two consecutive correction boluses. In such cases, patients were trained to act according to the protocol of hyperglycemia management by taking an insulin dose via conventional injection and changing the infusion set. The treating team members were available after 24 hours for all patient calls (dieticians were only available during the daytime).
Patients on MDI were also trained in carbohydrate counting and the use of insulin sensitivity factors for calculating correction boluses and had access to the treating team similar to the CSII group.
All statistical analysis were performed using SPSS Statistics (SPSS Inc., Chicago, USA) version 20. A p-value of less than 0.05 was considered statistically significant. Data were expressed as mean and standard deviation (SD) with 95% confidence intervals (CI). A Mann-Whitney U test was used to compare HbA1c between baseline and the last follow-up visit in both CSII and MDI groups. Student’s t-tests were used to compare the means of two normally distributed variables. Chi-square tests were used to compare proportions. The study was approved by the joint Faculty of Medicine and Ministry of Health Ethical Committee.
Results
The characteristics of 326 children on CSII and 328 on MDI are shown in Table 1. Mean age at entry, gender distribution, BMI, age at diagnosis, insulin dose, and duration of T1DM at entry were not significantly different between the two groups.
Table 1: Baseline characteristics of the study population using CSII and MDI.
|
Gender* |
|
Male |
125 (38.3) |
126 (38.4) |
|
|
Female |
201 (61.7) |
202 (61.6) |
|
|
Age at entry ( years) |
9.2±3.4 |
9.7±5.5 |
0.129 |
|
BMI (kg/m2) |
18.6±3.9 |
18.7±4.0 |
0.868 |
|
Age at diagnosis (years) |
6.2±2.9 |
6.2±3.0 |
0.938 |
|
Duration of diabetes at entry (years) |
5.5±2.8 |
5.4±2.7 |
0.134 |
|
Insulin dose
(U/kg/d) |
0.8±0.2 |
0.8±0.2 |
0.786 |
|
HbA1c (%) |
8.9±1.4 |
8.8±1.4 |
0.741 |
|
Reason for switching to CSII* |
|
For better control |
143 (43.9) |
|
|
|
Frequent hypoglycemia |
51 (15.6) |
|
|
|
Dislike/fear of needles |
49 (15) |
|
|
|
Better quality of life |
48 (14.7) |
|
|
|
Recurrent DKA |
19 (5.8) |
|
|
*n (%).
Data expressed as mean±SD.
CSII: continuous subcutaneous insulin infusion; MDI: multiple daily injections; BMI: body mass index; HBA1C: glycated hemoglobin;
DKA: diabetic ketoacidosis.
In the CSII cohort, 281 (86.0%), 211 (65.0%), 110 (33.7%), 53 (16.3%) and 27 (8.3%) subjects were followed for one, two, three, four, and five years, respectively. Only three patients (0.9%) discontinued the pump in the five-year study period. The government-funded program for insulin pumps was established in 2007, the number of patients who received pumps increased after that.
The main reasons for switching to CSII given by the patients or their caregivers were achieving better control (n=143; 43.9%) followed by frequent symptomatic hypoglycemia (n=51; 15.6%) [Table 1].
Figure 1 describes the percentage of patients achieving target HbA1c (≤7.5%). The numbers increased in the CSII group compared to MDI throughout the study period. There was no sex difference in achieving target HbA1c.
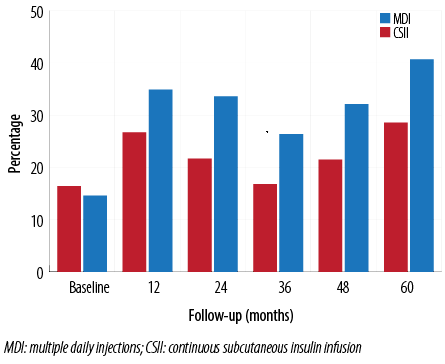
Figure 1: Percentage of children who achieved the target of HbA1c <7.5% at different time points during the five-year follow-up.
HbA1c dropped in both groups in the first year. After the second year HbA1c levels in the MDI group gradually increased again to values higher than those measured at baseline (8.9±1.7 at baseline vs. 9.0 ± 1.2 in the fifth year, p<0.001) [Figure 2]. The difference in the HbA1c values at baseline and in the fifth year in the CSII and MDI group were 8.9±1.4 and 8.3±1.2 vs. 8.8±1.4 and 9.0±1.6, respectively; p<0.05). There was no significant effect of sex on HbA1c values at baseline and throughout the follow-up period, except at the end fifth year (8.5±1.6 vs. 7.7±1.8 in males and females, respectively; p<0.001).
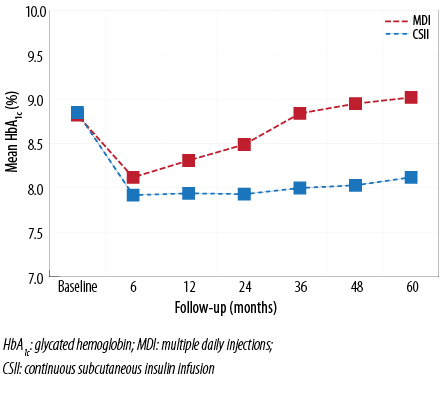
Figure 2: HbA1c levels in CSII and MDI groups from baseline to 60 months of follow-up. The drop in HbA1c was seen in both groups at six months; however, the drop was more significant in the CSII group throughout the follow-up period (p<0.001).
The mean age at CSII start was 9.2±3.4 years. HbA1c at pump initiation was higher in the youngest age group (8.9 vs. 8.3 vs. 8.7 in the <6, 6–12, and 13–18 year old groups, respectively; p<0.05). The improvement in HbA1c was most significant in the under 6-year-old group. At the end of the study period, the reduction from baseline was 15.7% in the youngest age group, 1.2% in the middle group, and 6.1% in the oldest age group [Figure 3].
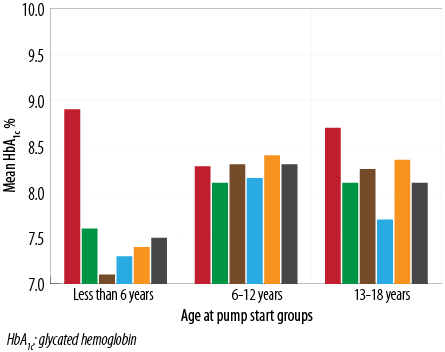
Figure 3: Mean HbA1c values at baseline and throughout the five-year follow-up period stratified by age at pump insertion.
The mean duration of T1DM at the start of the study was 5.5±2.8 years and 5.4±2.7 years in the MDI and CSII groups, respectively. We assessed the effect of disease duration on glycemic control. A positive relationship was observed between the T1DM duration at pump insertion and HbA1c improvement in the 3–5 year duration group. The change was consistent throughout the study period (9.0±1.3 at baseline and 8.4±2.1 at year five; p<0.001). Similar results were found in the group who had diabetes for more than five years (8.4±2.1 at baseline and 8.2±1.9 year five; p<0.01) [Figure 4].
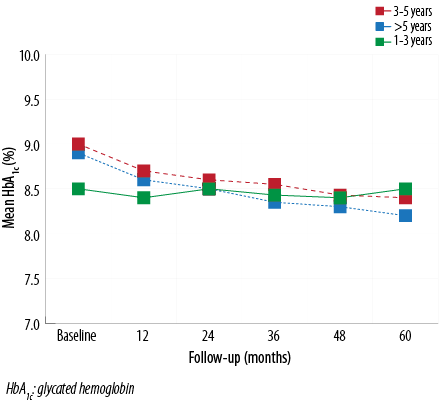
Figure 4: Mean HbA1c values at baseline and throughout the five-year follow-up period stratified by duration of diabetes (years). Those who had a duration of diabetes 3–5 years had the best HbA1c improvement throughout the follow-up period.
At baseline, the mean insulin doses were 0.9±0.3 and 0.9±0.3U/kg/d in the MDI and CSII groups, respectively (p=0.77). Compared to baseline, insulin requirements after CSII initiation decreased to 0.8±0.2 and 0.8±0.2U/kg/d in the MDI and CSII groups, respectively, in the first six months (p<0.001). At the end of the fifth year, the insulin requirement in the MDI group was higher than at baseline (0.9±0.3 vs. 0.9±0.3, p<0.05). In contrast, the dose at the same time point in the CSII group was significantly lower than at baseline (0.9±0.3 vs. 0.9±0.2, p<0.05) [Figure 5].
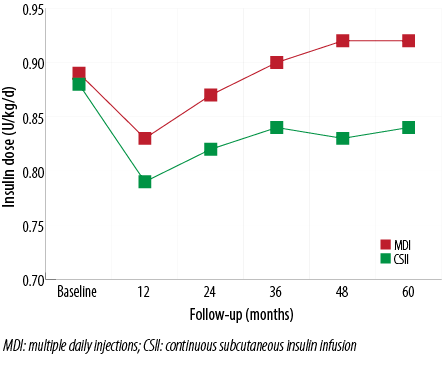
Figure 5: Daily insulin requirement changes in both CSII and MDI groups. After an initial decrease in the first year, insulin requirements in the MDI group rose progressively, eventually exceeding the doses at baseline at the end of the fifth year.
At baseline, BMI z-scores were similar in both groups and increased to similar degrees after the first year [Figure 6]. However, the increase was more significant in the CSII group (1.0 vs. 1.4, at the fifth year in the MDI and CSII groups, respectively; p<0.001). No significant difference was noted between males and females. Only three patients (two aged 16 and one aged 18 years) had mild elevation in their total cholesterol levels. Their triglycerides were normal. Two were in the CSII group and one in the MDI group. None had elevated blood pressure.
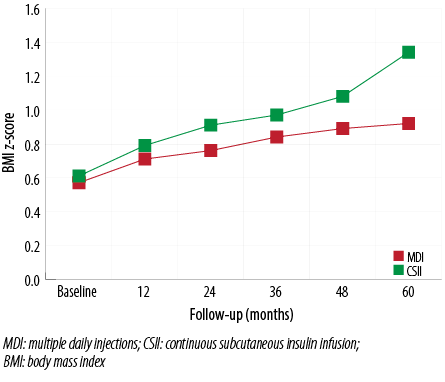
Figure 6: BMI changes in the CSII and MDI groups. Although the mean BMI z-scores increases were greater in the CSII group, the difference was statistically significant only at the end of the study.
Severe hypoglycemia decreased from 9.7 events/100-patient-years at baseline to 4.1 events/100-patient-years at the end of the follow-up period in the CSII group (p<0.05). However, it increased from 7.7 events/100-patient-years at baseline to 19.7 events/100-patient-years at the end of the study in the MDI group (p<0.05).
The rates of DKA episodes were not significantly different in the MDI and CSII groups at baseline (5.1 vs. 4.9 events/100-patient-years; p=0.08) or throughout the follow-up period (5.5 vs. 5.1, at the end of fifth year).
One patient on CSII and one on MDI developed lipoatrophy in the abdominal area, which was resolved after switching from insulin aspart to lispro. Mild skin irritation that required local treatment at the site of canula insertion developed in eight patients in the five-year follow-up. One patient developed infection requiring local antibiotics. Five patients had mild bruising. All events developed in the first three years of follow-up.
Discussion
This study represents the first analysis of CSII use in children and adolescents in Kuwait since the initiation of the government funded program in 2007. It compared the efficacy and safety of CSII in a large cohort of patients.
Our results clearly demonstrate a significant and persistent improvement in glycemic control (measured by HbA1c levels) in the CSII group compared to the MDI group. This is consistent with findings from other studies5,15-19 that showed significant reductions in HBA1c without increasing the risk for hypoglycemia. In a meta-analysis comparing CSII vs. MDI,20 HbA1c decreased more in patients treated with CSII in all trials. In a long-term study on a large number of children with T1DM,16 a sustained improvement in glycemic control was demonstrated in the CSII group compared to a matched cohort using injections. Furthermore, a recent retrospective, international, multicenter study by Mameli et al,21 associated the use of CSII with greater improvement in HbA1c after one year of treatment and during the seven-year follow-up.
Others also reported the slight increase in HbA1c after the initial drop seen in our study.17-19,21 In a three-year multicenter cohort analysis, Jakish19 showed the superiority of the CSII regime over MDI only in the first year; the difference did not persist. This could be attributed to reduced motivation and attention to the new treatment mode after the initial period.
In our study, there was no significant effect of sex on HbA1c levels except at the end of the fifth year of follow-up where females had lower levels. This was consistent with the finding of Shalitin,22 but not others21,23 who reported better glycemic control in males on CSII throughout the study period. The authors hypothesized that achieving glycemic control was more difficult in females due to their increased risk of depression and disturbed body image.
Children with pump initiation before the age of six had significantly better glycemic control compared to those who started at an older age. This was inconsistent with findings reported in other studies.20,24,25 Although the younger age group in our study had higher HbA1c at initiation, which has been shown to be associated with better improvement, the difference was significant throughout the entire five years of follow-up. One possible explanation could be the fact that this age group is under the “control” of the caregivers for blood glucose checking, carbohydrate counting, and pump data entry compared to the older age groups, for whom the influence of parents/caregivers is much less. Batajoo et al,26 and other researchers24,25,27 demonstrated a trend toward HbA1c improvement only in children greater than 12 years of age. On the other hand, the HbA1c in one study16 was not related to age at commencement of CSII therapy. This variation in the results could be explained by the differences in study design (pre- and post-pump, cross-over and MDI vs. CSII).
A shorter duration of T1DM before pump initiation has been associated with achievement of target HbA1c.28 However, in our study and one other,29 a better response was observed in the group with longer T1DM duration before pump initiation (3–5 years and >5 years). Possible reasons for the variability between our results and those previously reported may be the differences in study designs and the duration between T1DM onset and initiation of pump therapy (ranging from a few weeks to years).
Patients in the CSII group required less daily insulin throughout the study period. This observation was reported in many short- and long-term studies.16,18-20,22,27,30 Insulin delivery by CSII more closely mimics physiological insulin secretion and, consequently, less insulin is needed to achieve glycemic control compared to MDI. Although there was an increase in insulin requirement after the second year, it remained lower than the baseline value throughout the study period. This may be a positive factor when assessing treatment cost.
Intensive insulin therapy has been reported to cause weight gain.2 However, the data regarding insulin pump therapy are conflicting. Some studies, like ours, have demonstrated an increase in weight after CSII initiation11,16,25,31,32 despite the reduction in daily insulin dose. The increased prevalence of overweight and obesity in children in the Middle East, including Kuwait,33 and the feeling of “freedom” to eat without extra injections may have contributed to the weight gain in the CSII group in our study. On the other hand, others have shown that average BMI z-scores gradually deceased18,34,35 or remained unchanged.21,23,36 Lower insulin requirements and intensive nutritional education with CSII therapy could explain this observation.
Although insulin pump therapy improved glycemic control, this was not associated with an increased risk of severe hypoglycemia. In fact, there was a significant reduction in hypoglycemia in the CSII group compared to MDI throughout the study period. This observation was consistent with many earlier studies,9-11,13,18,23,25,27,31,37 even when there was no improvement in HbA1c.27 This could be explained by the fact that insulin delivery by insulin pump is more physiologic, and the availability of multiple basal rates based on activity, sleeping, and eating patterns decreases the frequency of severe hypoglycemic events. There was no significant difference in the rates of DKA (events/100-patient-years) between the CSII and MDI groups in our study, although there was a trend towards fewer events in the CSII. Earlier studies reported similar finding.20,24,30,32,35 Conversely, Shalitin22 reported an increase in DKA episodes from 0.03 to 0.07 events/100 patient-years. Most of them were caused by technical faults interrupting insulin delivery and delay in response from patient/caregivers.
The strength of our study was the large patient numbers in the CSII group and the long follow-up period. Although patients on MDI were used as a control group, the main limitation of the study is the lack of randomization between the CSII and MDI group. Moreover, we did not evaluate the number of clinic visits for MDI and CSII group, which has been shown to affect glycemic control. However, our patients were seen every 4–6 weeks (after the initial weekly period) regardless of their treatment modality and all received the same education with the same educators. They also had 24-hour access to the treatment teams.
Conclusion
Insulin pump therapy is an effective and safe mode of insulin delivery system in children and adolescents with T1DM. The use of advanced features of the pump and the continuous glucose monitoring system will benefit glycemic control and reduce the frequency and duration of hypoglycemia. Special attention and education regarding healthy eating habits is mandatory to avoid weight gain. Active involvement of the patients and their caregivers, accompanied with support from the medical team, is needed to maximize the benefits of insulin pumps. Cost-effectiveness needs to be evaluated especially in areas with limited resources.
Disclosure
The authors declared no conflicts of interest. The research was funded by the Kuwait University Research Department (MK 02/13).
references
- Diabetes Atlas-IDF. International Diabetes Federation, 4th Edition. 2013.
- The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 1993 Sep;329(14):977-986.
- Kordonouri O, Hartmann R, Danne T. Treatment of type 1 diabetes in children and adolescents using modern insulin pumps. Diabetes Res Clin Pract 2011 Aug;93(Suppl 1):S118-S124.
- Skinner TC, Cameron FJ. Improving glycaemic control in children and adolescents: which aspects of therapy really matter? Diabet Med 2010 Apr;27(4):369-375.
- Weissberg-Benchell J, Antisdel-Lomaglio J, Seshadri R. Insulin pump therapy: a meta-analysis. Diabetes Care 2003 Apr;26(4):1079-1087.
- Weintrob N, Benzaquen H, Galatzer A, Shalitin S, Lazar L, Fayman G, et al. Comparison of continuous subcutaneous insulin infusion and multiple daily injection regimens in children with type 1 diabetes: a randomized open crossover trial. Pediatrics 2003 Sep;112(3 Pt 1):559-564.
- DiMeglio LA, Pottorff TM, Boyd SR, France L, Fineberg N, Eugster EA. A randomized, controlled study of insulin pump therapy in diabetic preschoolers. J Pediatr 2004 Sep;145(3):380-384.
- Pickup J, Mattock M, Kerry S. Glycaemic control with continuous subcutaneous insulin infusion compared with intensive insulin injections in patients with type 1 diabetes: meta-analysis of randomised controlled trials. BMJ 2002 Mar;324(7339):705.
- Boland EA, Grey M, Oesterle A, Fredrickson L, Tamborlane WV. Continuous subcutaneous insulin infusion. A new way to lower risk of severe hypoglycemia, improve metabolic control, and enhance coping in adolescents with type 1 diabetes. Diabetes Care 1999 Nov;22(11):1779-1784.
- Plotnick LP, Clark LM, Brancati FL, Erlinger T. Safety and effectiveness of insulin pump therapy in children and adolescents with type 1 diabetes. Diabetes Care 2003 Apr;26(4):1142-1146.
- Battelino T. Risk and benefits of continuous subcutaneous insulin infusion (CSII) treatment in school children and adolescents. Pediatr Diabetes 2006 Aug;7(Suppl 4):20-24.
- Hanas R, Lindgren F, Lindblad B. A 2-yr national population study of pediatric ketoacidosis in Sweden: predisposing conditions and insulin pump use. Pediatr Diabetes 2009 Feb;10(1):33-37.
- Shaltout A, Abdulrasoul M, AlKandari H, AlMahdi M, AlKhawari M. Incidence of type 1 diabetes in children and adolescents (0-19 years) in Kuwait. National Diabetes Registry Data. (Unpublished Data)
- Global IDF/ISPAD Guideline for Diabetes in childhood and Adolescents. International Diabetes Federation, 2011; 9-15.
- Weinzimer SA, Swan KL, Sikes KA, Ahern JH. Emerging evidence for the use of insulin pump therapy in infants, toddlers, and preschool-aged children with type 1 diabetes. Pediatr Diabetes 2006 Aug;7(Suppl 4):15-19.
- Johnson SR, Cooper MN, Jones TW, Davis EA. Long-term outcome of insulin pump therapy in children with type 1 diabetes assessed in a large population-based case-control study. Diabetologia 2013 Nov;56(11):2392-2400.
- Pickup J, Keen H. Continuous subcutaneous insulin infusion at 25 years: evidence base for the expanding use of insulin pump therapy in type 1 diabetes. Diabetes Care 2002 Mar;25(3):593-598.
- Sulli N, Shashaj B. Long-term benefits of continuous subcutaneous insulin infusion in children with Type 1 diabetes: a 4-year follow-up. Diabet Med 2006 Aug;23(8):900-906.
- Jakisch BI, Wagner VM, Heidtmann B, Lepler R, Holterhus PM, Kapellen TM, et al; German/Austrian DPV Initiative and Working Group for Paediatric Pump Therapy. Comparison of continuous subcutaneous insulin infusion (CSII) and multiple daily injections (MDI) in paediatric Type 1 diabetes: a multicentre matched-pair cohort analysis over 3 years. Diabet Med 2008 Jan;25(1):80-85.
- Jeitler K, Horvath K, Berghold A, Gratzer TW, Neeser K, Pieber TR, et al. Continuous subcutaneous insulin infusion versus multiple daily insulin injections in patients with diabetes mellitus: systematic review and meta-analysis. Diabetologia 2008 Jun;51(6):941-951.
- Mameli C, Scaramuzza AE, Ho J, Cardona-Hernandez R, Suarez-Ortega L, Zuccotti GV. A 7-year follow-up retrospective, international, multicenter study of insulin pump therapy in children and adolescents with type 1 diabetes. Acta Diabetol 2014 Apr;51(2):205-210.
- Shalitin S, Gil M, Nimri R, de Vries L, Gavan MY, Phillip M. Predictors of glycaemic control in patients with Type 1 diabetes commencing continuous subcutaneous insulin infusion therapy. Diabet Med 2010 Mar;27(3):339-347.
- Deiss D, Hartmann R, Hoeffe J, Kordonouri O. Assessment of glycemic control by continuous glucose monitoring system in 50 children with type 1 diabetes starting on insulin pump therapy. Pediatr Diabetes 2004 Sep;5(3):117-121.
- Doyle EA, Weinzimer SA, Steffen AT, Ahern JA, Vincent M, Tamborlane WV. A randomized, prospective trial comparing the efficacy of continuous subcutaneous insulin infusion with multiple daily injections using insulin glargine. Diabetes Care 2004 Jul;27(7):1554-1558.
- Ahern JA, Boland EA, Doane R, Ahern JJ, Rose P, Vincent M, et al. Insulin pump therapy in pediatrics: a therapeutic alternative to safely lower HbA1c levels across all age groups. Pediatr Diabetes 2002 Mar;3(1):10-15.
- Batajoo RJ, Messina CR, Wilson TA. Long-term efficacy of insulin pump therapy in children with type 1 diabetes mellitus. J Clin Res Pediatr Endocrinol 2012 Sep;4(3):127-131.
- Sulli N, Shashaj B. Continuous subcutaneous insulin infusion in children and adolescents with diabetes mellitus: decreased HbA1c with low risk of hypoglycemia. J Pediatr Endocrinol Metab 2003 Mar;16(3):393-399.
- Shalitin S, Gil M, Nimri R, de Vries L, Gavan MY, Phillip M. Predictors of glycaemic control in patients with Type 1 diabetes commencing continuous subcutaneous insulin infusion therapy. Diabet Med 2010 Mar;27(3):339-347.
- Pinhas-Hamiel O, Tzadok M, Hirsh G, Boyko V, Graph-Barel C, Lerner-Geva L, et al. The impact of baseline hemoglobin A1c levels prior to initiation of pump therapy on long-term metabolic control. Diabetes Technol Ther 2010 Jul;12(7):567-573.
- Johannesen J, Eising S, Kohlwes S, Riis S, Beck M, Carstensen B, et al. Treatment of Danish adolescent diabetic patients with CSII - a matched study to MDI. Pediatr Diabetes 2008 Feb;9(1):23-28.
- Hathout EH, McClintock T, Sharkey J, Kytsenko D, Hartwick N, Hadley-Scofield M, et al. Glycemic, auxologic, and seasonal aspects of continuous subcutaneous insulin infusion therapy in children and young adults with type 1 diabetes. Diabetes Technol Ther 2003;5(2):175-181.
- Fendler W, Baranowska AI, Mianowska B, Szadkowska A, Mlynarski W. Three-year comparison of subcutaneous insulin pump treatment with multi-daily injections on HbA1c, its variability and hospital burden of children with type 1 diabetes. Acta Diabetol 2012 Oct;49(5):363-370.
- Ministry of Health. Kuwait. Administration of Food and Nutrition, 2013. Kuwait Nutrition Surveillance System 2007-2013 report.
- Raile K, Noelle V, Landgraf R, Schwarz HP. Weight in adolescents with type 1 diabetes mellitus during continuous subcutaneous insulin infusion (CSII) therapy. J Pediatr Endocrinol Metab 2002 May;15(5):607-612.
- Willi SM, Planton J, Egede L, Schwarz S. Benefits of continuous subcutaneous insulin infusion in children with type 1 diabetes. J Pediatr 2003 Dec;143(6):796-801.
- Bin-Abbas BS, Sakati NA, Raef H, Al-Ashwal AA. Continuous subcutaneous insulin infusion in type 1 diabetic Saudi children. A comparison with conventional insulin therapy. Saudi Med J 2005 Jun;26(6):918-922.
- Karagianni P, Sampanis Ch, Katsoulis Ch, Miserlis G, Polyzos S, Zografou I, et al. Continuous subcutaneous insulin infusion versus multiple daily injections. Hippokratia 2009 Apr;13(2):93-96.