Vascular complications post-renal transplantation are uncommon but represent a serious problem.1 Renal artery aneurysms constitute less than 1% of all post-transplant vascular complications. They are devastating as they may cause functional impairment or loss of the graft.1–5 Indications for repair include symptomatic aneurysms, size larger than 2.5 cm, the presence of infection, and the progressive increase in size that precipitates a life-threatening rupture.1,3,4,6,7 Therapeutic options include open surgical repair, ultrasound guided percutaneous administration of thrombin, and endovascular repair.1,4,6
We report a case of a transplanted kidney renal artery aneurysm that was treated using endovascular repair with successful preservation of the allograft.
Case report
A 35-year-old man underwent a living donor kidney transplant at our transplantation unit in 2003. His postoperative course was uneventful. The procedure resulted in normalization of his renal function and no signs of rejection were reported in his follow-up records.
The patient was compliant with his immune-suppressive therapy and maintained frequent follow-up visits to his nephrologist during which he had normal renal transplant function.
In 2013, the patient travelled outside the country for work purposes. During his overseas stay, he had gradual deterioration in his kidney function for which he was treated conservatively until his creatinine level ranged between 2.0–3.0 mg/dL. A kidney biopsy of the transplant was performed in March 2014 and he was informed that no obvious pathology could be determined. Conservative management was continued.
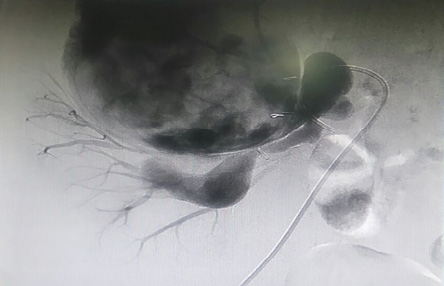
Figure 1: Selective angiogram showing the upper renal artery with the proximal branches supplying the kidney, and the dilated distal segment with the aneurysm arising from it.
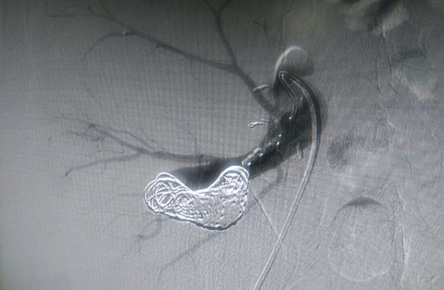
Figure 2: Completion angiogram showing preservation of proximal branches of the upper renal artery and no contrast filling the aneurysm.
In June 2015, the patient returned home and he was admitted for further investigations at our transplant unit. Upon admission, a right iliac fossa pulsatile mildly tender mass was felt and a bruit was clearly audible. His creatinine ranged between 4.0–4.5 mg/dL. Duplex ultrasound revealed a 110 × 90 mm partially thrombosed aneurysm adjacent to the upper half of the transplanted kidney. The exact origin of the aneurysm could not be determined by the ultrasound. A multisliced computed tomography (CT) angiogram showed a 115 × 91 mm false aneurysm extending superiorly to a level above the aortic bifurcation.
A three-dimensional reconstruction of the CT scan demonstrated that the transplanted kidney was supplied by two arteries. The aneurysm was originating from the distal part of the upper renal artery (URA), which was anastomosed to the common iliac artery during the original surgery. Few branches were identified originating from the proximal part of URA and supplying parts of the upper and middle segments of the kidney.
The lower pole of the kidney was supplied by the lower renal artery (LRA) that was anastomosed to the external iliac artery during the original surgery. This artery was patent and within normal diameter.
Based on having a huge aneurysm and a favorable anatomy, the patient was offered an endovascular repair. Informed consent was obtained after the risk of graft loss and the possibility to convert to surgical intervention with an associated risk of nephrectomy were explained to the patient. Blood culture was negative.
Using a left common femoral artery puncture, a selective angiogram of the URA was performed. The proximal segment, about 3 cm in length, appeared normal. Three branches were originating from this proximal segment and supplying the upper and middle parts of the kidney. The distal segment of the URA was dilated and tortuous to 2.5 cm followed by a significant narrowing which was 4–5 mm in diameter and 25 mm in length after which the contrast filled the huge aneurysm [Figure 1].
Two MVP-5 plugs (Reverse Medical Corp, micro vascular plug system, Irvine, CA) were applied to the most distal narrowed segment of the URA resulting in total occlusion. The dilated and tortuous segment of the URA was filled with 2 mL of liquid embolic system (PHIL 30%, Microvention, Inc California, USA) and four Axium™ coils (ev3, Irvine, CA, USA) sizes 18 mm × 40 cm and 20 mm × 50 cm. Completion angiogram revealed preservation of the three proximal branches of the URA with no contrast filling the aneurysm and a patent LRA [Figure 2].
Postoperative course was uneventful. His serum creatinine dropped progressively to reach 2.6 mg/dL on the fourth postoperative day during which he was discharged. At one-year follow-up, the patient’s kidney function stabilized within a range of 2.2–2.5 mg/dL and the duplex ultrasound continued to show a thrombosed aneurysm with decrease in its size to 100 × 74 mm.
Discussion
Vascular complications post kidney transplant are not common but result in a high rate of allograft loss.1 The incidence of vascular complications has been reported in different studies to range between 3–14%. Among these complications are renal artery stenosis, renal artery thrombosis, renal vein thrombosis, arteriovenous fistula, and renal artery aneurysm.5,6,8 Although transplanted renal artery aneurysms are rare constituting 0.5–1.0% of all vascular complications, they are devastating as functional impairment and nephrectomy are likely in these cases.1, 2–5
Despite renal artery aneurysms being mostly asymptomatic, some patients still may present with fever, prolonged anemia, ischemia of the ipsilateral limb, hypertension, compression of adjacent organs, deterioration in graft function, and life-threatening bleeding due to rupture.1–3,7 Our patient’s aneurysm was diagnosed while investigating the gradual deterioration in his kidney function test.
Although renal artery aneurysms mostly occur in the early stages post-transplantation, they can occasionally occur as late complications as in our case where the patient presented 12 years after his transplantation.4, 6
In the few cases of transplanted renal artery aneurysms that were reported in the literature,7 several factors were included in the etiology. A defective surgical technique with poor suturing, arterial kinking and instrumental injury can lead to suture rupture with anastomotic leakage and extra-renal aneurysm formation.1,7 Also, perivascular infection may cause vessel wall ischemia and arterial anastomotic dehiscence.1,7
Immunological factors such as acute or chronic rejection were suggested in some reports, especially in cases of intrarenal false aneurysms. However, this remains a hypothesis and needs to be proven.7
Our patient’s aneurysm was not anastomotic as it was identified few centimeters from the suture line and the blood culture was negative. Although the report was lacking, a recent renal biopsy can be added as a possible iatrogenic cause of the aneurysm.
Although conventional angiogram is the gold standard modality for diagnosis of renal artery aneurysms, color flow duplex scanning and multisliced CT angiogram are of great value in the diagnosis.1,4 Due to the impaired renal function of our patient, we chose to start with duplex scanning which detected the aneurysm but could not locate the site of origin. We proceeded with CT angiogram to clarify the anatomy and select the best possible treatment option.
Studies advocate the following indications for intervention: symptomatic aneurysms, a size larger than 2.5 cm, if complicated by infection, progressive enlargement with impending risk of rupture.1,3,4,6,7 Asymptomatic small aneurysms can be managed conservatively with close surveillance. In our report, the indication for treatment was the huge size of the aneurysm with the associated high risk of rupture.
Current therapeutic options include open repair, endovascular repair, and ultrasound guided percutaneous thrombin injection.1,2,4,6 Open surgical repair includes resection of the aneurysm and arterial reconstruction using patch angioplasty, reanastomosis, or allograft autotransplantation.1,2,7 Endovascular repair is another valuable treatment option with the deployment of a covered stent to exclude the renal transplant artery in cases of rupture with subsequent nephrectomy.5,6 Also in cases of sufficient proximal and distal landing zones, a stent can be used to exclude the aneurysm. Ultrasound guided injection of thrombin was also applied in cases where the aneurysm was accessible and in the absence of infection.6
In our patient, several proximal branches originated from the URA supplying the upper half of the kidney. Additionally, an adequate length of this artery distal to its proximal branches was available making the endovascular intervention a suitable solution for the exclusion of this huge aneurysm.
As stated in other case reports, the length of hospital stay post-endovascular repair was shorter than the stay after the open repair.7 Similarly for our patient, the length of the hospital stay was relatively short as he was discharged on the fourth day.
Conclusion
Renal artery aneurysm in a transplanted kidney is rare, but serious complication may lead to allograft loss. Open surgical repair, thrombin injection, and endovascular repair are valid modalities of treatment. With proper selection, we believe that endovascular treatment provides a safe and less invasive modality with a shorter length of hospital stay.
Disclosure
The authors declared no conflicts of interest.
references
- 1. Al-Wahaibi KN, Aquil S, Al-Sukaiti R, Al-Riyami D, Al-Busaidi Q. Transplant renal artery false aneurysm: case report and literature review. Oman Med J 2010 Oct;25(4):306-310.
- 2. Amirzargar MA, Ilati AA, Drakhshanfar A, Poorolajal J, Eslami A. Internal iliac artery aneurysm resection in transplanted kidney. Arch Iran Med 2014 Dec;17(12):844-846.
- 3. Busato CR, Utrabo CA, de Sousa WF, Gomes RZ, Hosoume JK, Hoeldtke E, et al. Renal artery aneurysm in a transplanted kidney: ex vivo graft repair and reimplantation. J Vasc Bras 2009 Jan/Mar;8(1).
- 4. Bracale UM, Santangelo M, Carbone F, Del Guercio L, Maurea S, Porcellini M, et al. Anastomotic pseudoaneurysm complicating renal transplantation: treatment options. Eur J Vasc Endovasc Surg 2010 May;39(5):565-568.
- 5. Gang S, Rajapurkar M. Vascular complications following renal transplantation. J Nephrol Ren Transplant 2009;2(1):122-132.
- 6. Buimer MG, van Hamersvelt HW, Adam van der Vliet J. Anastomotic pseudoaneurysm after renal transplantation; a new hybrid approach with graft salvage. Transpl Int 2012 Jul;25(7):e86-e88.
- 7. Bracale UM, Carbone F, del Guercio L, Viola D, D’Armiento FP, Maurea S, et al. External iliac artery pseudoaneurysm complicating renal transplantation. Interact Cardiovasc Thorac Surg 2009 Jun;8(6):654-660.
- 8. Dimitroulis D, Bokos J, Zavos G, Nikiteas N, Karidis NP, Katsaronis P, et al. Vascular complications in renal transplantation: a single-center experience in 1367 renal transplantations and review of the literature. Transplant Proc 2009 Jun;41(5):1609-1614.