Dyke-Davidoff-Masson syndrome (DDMS) is characterized by the presence of hemiatrophy of one cerebral hemisphere, which is usually a consequence of an insult to the developing brain. This condition is clinically characterized by facial asymmetry, contralateral spastic hemiplegia or hemiparesis, mental retardation, and refractory seizure. A refractory seizure is usually the presenting feature of this condition and often poses a therapeutic challenge to the treating physician.
The radiological features of DDMS are distinctive, and may include cerebral hemiatrophy with several parenchymal changes and ipsilateral compensatory hypertrophy of the skull bone and sinuses.1,2
We describe three cases of patients with DDMS, who initially presented with a refractory seizure. In this report, we also seek to emphasize the importance of thorough neuroimaging of any child with seizure disorder to obtain the appropriate diagnosis.
Case Reports
Case one
A 12-year-old boy presented to the pediatric department of North Bengal Medical College and Hospital for the evaluation of repeated partial seizures involving the right half of his body for the last five years.
He was delivered at home after an uneventful antenatal period. There was no complication in the postnatal period. According to his mother, he showed developmental delay since early infancy. At the age of seven, he started to have repeated episodes of convulsion involving the right half of the body despite institution of multiple antiepileptic drugs (phenytoin, levetiracetam, and clobazam). On examination, there was comparative shortening of the right upper and lower limbs [Figure 1]. He had slurred speech, spastic hemiparesis with brisk tendon reflexes, and extensor plantar response of the right side. There was also mild contracture of the right tendo Achilles. Cranial nerves examination was normal.
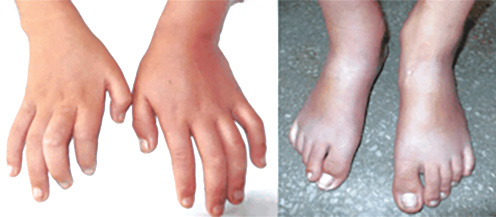
Figure 1: Photograph showing comparative shorting of the right hand and foot.
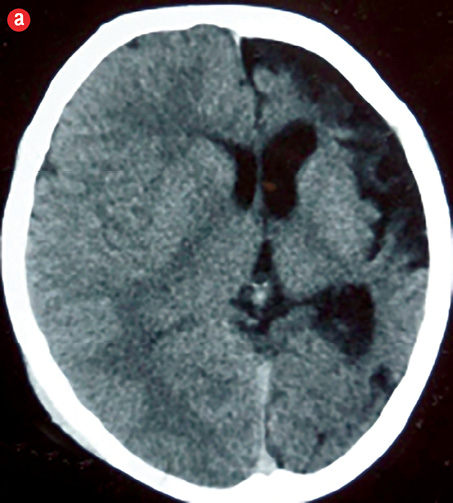
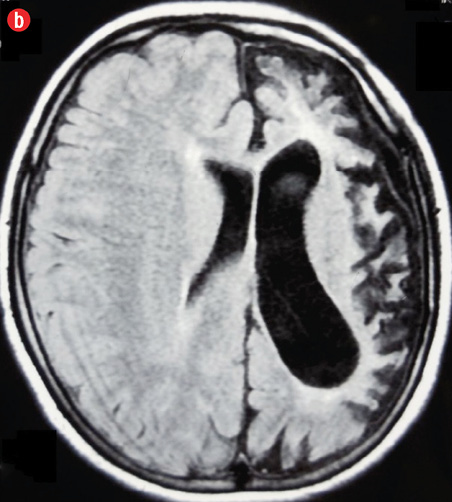
A computed tomography (CT) scan of the head revealed atrophy of the left cerebral hemisphere with dilatation of the ipsilateral ventricle and widening of the sulci and Sylvian fissure of the left side. Extra-axial cerebrospinal fluid (CSF) spaces over the left cerebral convexity were prominent, and there was a shifting of the midline falx towards the left side. Thickening of the calvarial bones, especially the left squamous of the temporal bone was also noted. Additionally, magnetic resonance imaging (MRI) of the brain showed small left cerebral hemisphere with prominent sulci and Sylvian fissure, enlargement of left-sided frontal sinus, elevation of left petrous ridge, and hypoplasia of left-sided cerebral peduncle [Figure 2 and 3]. Based on the clinical and characteristic imaging, a diagnosis of DDMS was made.
|
|
|
|
|
Figure 2: (a) Non-contrast computed tomography and (b) magnetic resonance imaging (T1-weighted) showing left cerebral hemiatrophy with prominent ipsilateral ventricle, sulci, and Sylvian fissure. Extra-axial cerebrospinal fluid spaces are prominent over the left cerebral convexity and shifting of midline falx towards left side. |
Figure 3: (a and b) Coronal magnetic resonance imaging (T2-weighted), and (c) axial non-contrast computed tomography scan showing prominent left frontal sinus (a; arrow), elevation of left petrous (b; arrow), and thickening of left squamous of the temporal bone (c; arrow).
Case two
A nine-year-old boy presented with a history of repeated attacks of right sided focal motor seizure followed by secondary generalization. Surprisingly, the child was on antiepileptic drugs for a long duration, but no neuroimaging was done. His birth and perinatal history was uneventful. The child had an asymmetrical face, delayed developmental milestones, and was mentally retarded. The clinical and CT scan findings were suggestive for the diagnosis of DDMS [Table 1].
Table 1: Distribution of cases clinical and radiological features.
|
1 |
12 |
Male |
Refractory hemiconvulsions |
rt.hemiatrophy rt.hemiparesis, UMN sign++, rt.tendo Achilles contracture, MR |
Left |
Cerebral atrophy, prominent sulci, prominent ventricle midline shift, bony thickening |
Prominent left frontal sinus, cerebral peduncle hypoplasia, and elevation of left petrous ridge |
|
2 |
9 |
Male |
rt.focal and GTC |
Asymmetrical face, MR, behavioral problem, learning disability, rt.hemiparesis, UMN sign+, |
Left |
Cerebral atrophy, prominent ventricle, widening of sulci and Sylvian fissure, bony thickening |
NA |
rt: right; UMN: upper motor neuron; MR: mental retardation; NA: not available; GTC: generalized tonic-clonic; CT: computed tomography; MRI: magnetic resonance imaging.
Case three
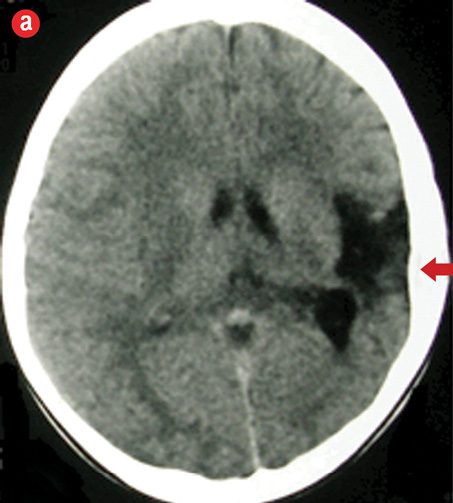
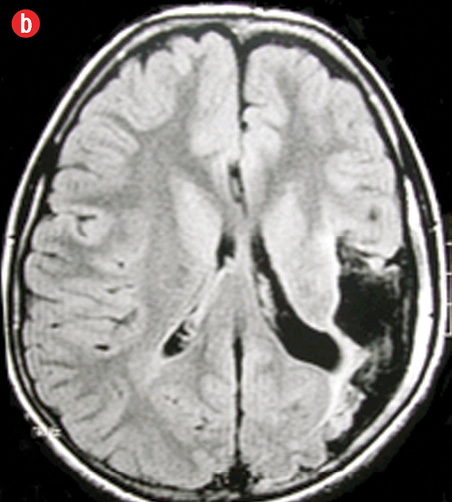
A 10-year-old boy, initially diagnosed with primary epilepsy based on electroencephalogram (EEG) findings, was on escalating doses of antiepileptic drugs since the age of three. He was admitted to our institution with refractory seizure, which needed an additional three anticonvulsants to control. His mother gave no history of perinatal asphyxia but noted his delayed developmental milestones. Clinical and neuroimaging findings clinched the diagnosis of DDMS [Figure 4].
|
|
|
|
|
Figure 4: (a) Non-contrast computed tomography showing left cerebral hemiatrophy with prominent ipsilateral ventricle, sulci, Sylvian fissure, and thickening of left sided calvarium (arrow). (b) magnetic resonance imaging (T1-weighted) revealed gliosis in left parieto-occipital cortex. |
All three cases presented around 10 years of age with refractory seizure, and diagnosis of DDMS was feasible after neuroimaging of the brain was done. In all cases, maternal antenatal history was uneventful and there was no history of any incident leading to postnatal brain insult. Besides neurological findings, other systemic examination was non-contributory, and laboratory parameters were within normal limits. Patients were counseled about the disease and its multidisciplinary management. The cases have been summarized in Table 1.
Discussion
DDMS was first described by Dyke, Davidoff, and Masson in 1933, when they described the plain skull radiographic and pneumoencephalographic changes in a series of nine patients.3 Both genders and any part of the hemisphere may be affected, but male sex and left hemisphere involvement are more frequent, as was also evident in our cases.4,5 The clinical features of this condition are variable, and depend on the onset and the extent of brain injury. Varying degrees of atrophy of one-half of the body, sensory loss, speech and language disorder, mental retardation or learning disability, and psychiatric manifestations have been reported.2,5
DDMS is classified into congenital and acquired type. The congenital variety mainly occurs due to in utero vascular occlusion involving the middle cerebral artery, congenital malformations, perinatal hypoxia, and infection. The main etiologic factors of acquired variety may include infection, trauma, tumor, ischemia, and hemorrhage. Age of presentation depends on the time of vascular occlusion of the middle cerebral vascular territory. The hemiatrophic cerebral parenchyma will have prominent sulci if the vascular insult occurs after birth, or after sulcation is completed. Whereas, if the vascular ischemia occurs during embryogenesis, no prominent sulci will be present [Table 2].1,6,7 A series of 19 cases of DDMS showed 12 cases as congenital and seven cases as acquired type.5 In our series, all three cases were likely to be of the congenital variety.
Table 2: Etiopathogenesis of Dyke-Davidoff-Masson syndrome.
|
Etiology |
In utero vascular occlusion involving the middle cerebral artery, congenital malformations perinatal hypoxia, and infection |
Infection, trauma, ischemia, tumor, hemorrhage |
|
Pattern of sulci |
Sulci may not be prominent |
Prominent sulci |
Besides cerebral hemiatrophy, other probable brain parenchymal changes are hypoplasia of mesencephalon, lentiform nucleus and thalamus, diffuse cerebellar atrophy, and ipsilateral atrophy of the cerebral peduncle.1,5 Hypoperfusion and decreased metabolic activity of the involved atrophied cerebral hemisphere have been demonstrated by positron emission tomography (PET).8,9
When brain damage occurs before the age of two, compensatory skull changes take place as an adaptation to the unilateral decrease of brain parenchymal volume. These compensatory changes include ipsilateral calvarial thickening, hyper pneumatization of the ipsilateral paranasal sinuses, and enlargement of mastoid cells. Elevation of the petrous ridge, sphenoid wing and orbital roof, hypoplasia of the anterior or middle cranial fossa, and shifting of the midline structures towards the atrophic hemisphere may be noted. These characteristic changes are seen in the congenital type compared to the acquired variety, and usually become evident in adolescence.1,6,7
The majority (60–70%) of patients with DDMS present with a generalized seizure disorder, and the rest present with focal seizure, or complex partial seizure with secondary generalization. Multiple anticonvulsants are usually required to control the seizures of these patients.4,5,10
Differential diagnosis to be considered in a patient with cerebral hemiatrophy include Sturge-Weber syndrome, Rasmussen encephalitis, Silver-Russel syndrome, and basal ganglia germinoma. Conditions associated with unilateral megalencephaly, as in the linear nevus syndrome, should also be considered.1,6,7
The typical features of Sturge-Weber syndrome include port-wine facial nevus, associated with leptomeningeal angioma intracranial tram track calcification, and cerebral atrophy. Rasmussen encephalitis is an immune mediated brain disorder leading to unilateral hemispheric atrophy, intractable focal epilepsy, and progressive neurological dysfunction. The imaging features comprise atrophy of unihemispheric focal cortical and ipsilateral head of caudate nucleus, without any calvarial changes.11 Hemiconvulsion-hemiplegia-epilepsy (HHE) syndrome should also be considered a clinical differential diagnosis of DDMS. HHE is characterized by the occurrence of prolonged clonic seizures with unilateral predominance occurring in the course of a febrile illness in a younger child and followed by the development of hemiplegia.12
The treatment of DDMS is symptomatic, typically using anticonvulsants and managing hemiplegia or hemiparesis, and addressing learning disability. Prognosis is better for patients who develop hemiparesis after the age of two years and in the absence of prolonged or recurrent seizures. Children with intractable disabling seizure and hemiplegia may benefit from hemispherectomy.9
Conclusion
DDMS should be considered as a differential diagnosis of refractory seizures in children. Along with a detailed history and complete clinical examination, neuroimaging imperative for correct diagnosis and proper management of such patients.
Disclosure
The authors declared no conflicts of interest.
references
- 1. Aguiar PH, Liu CW, Leitão H, Issa F, Lepski G, Figueiredo EG, et al. MR and CT imaging in the Dyke-Davidoff-Masson syndrome. Report of three cases and contribution to pathogenesis and differential diagnosis. Arq Neuropsiquiatr 1998 Dec;56(4):803-807.
- 2. Park KI, Chung JM, Kim JY. Dyke-Davidoff-Masson syndrome: cases of two brothers and literature review. J Epilepsy Res 2014 Jun;4(1):24-27.
- 3. Dyke CG, Davidoff LM, Masson CB. Cerebral hemiatrophy and homolateral hypertrophy of the skull and sinuses. Surg Gynecol Obstet 1933;57:588-600.
- 4. Unal O, Tombul T, Cirak B, Anlar O, Incesu L, Kayan M. Left hemisphere and male sex dominance of cerebral hemiatrophy (Dyke-Davidoff-Masson Syndrome). Clin Imaging 2004 May-Jun;28(3):163-165.
- 5. Atalar MH, Icagasioglu D, Tas F. Cerebral hemiatrophy (Dyke-Davidoff-Masson syndrome) in childhood: clinicoradiological analysis of 19 cases. Pediatr Int 2007 Feb;49(1):70-75.
- 6. Uduma FU, Emejulu JK, Motah M, Okere PC, Ongolo PC, Muna W. Differential diagnoses of cerebral hemiatrophy in childhood: a review of literature with illustrative report of two cases. Glob J Health Sci 2013 May;5(3):195-207.
- 7. Arora R, Rani JY. Dyke-Davidoff-Masson syndrome: imaging features with illustration of two cases. Quant Imaging Med Surg 2015 Jun;5(3):469-471.
- 8. Demir Y, Sürücü E, Çilingir V, Bulut MD, Tombul T. Dyke-Davidoff-Masson Syndrome With Cerebral Hypometabolism and Unique Crossed Cerebellar Diaschisis in 18F-FDG PET/CT. Clin Nucl Med 2015 Sep;40(9):757-758.
- 9. Shrestha B. Acquired cerebral hemiatrophy: Dyke-Davidoff-Masson Syndrome - a case report. Turk Neurosurg 2013;23(1):117-121.
- 10. Hsin YL, Chuang MF, Shen TW, Harnod T. Temporo-spatial analysis define epileptogenic and functional zone in a case of DDMS. Seizure 2011;20:713-716.
- 11. Varghese B, Aneesh M, Singh N, Gilwaz P. A case of Rasmussen encephalitis: the differential diagnoses and role of diagnostic imaging. Oman Med J 2014 Jan;29(1):67-70.
- 12. Auvin S, Bellavoine V, Merdariu D, Delanoë C, Elmaleh-Bergés M, Gressens P, et al. Hemiconvulsion-hemiplegia-epilepsy syndrome: current understandings. Eur J Paediatr Neurol 2012 Sep;16(5):413-421.