| |
1Departments of Anatomy, K.M.C.T. Medical College, Calicut University, Calicut, India 2Department of Anatomy, Kasturba Medical College, Manipal University, Mangalore, India. 3Department of Anatomy, St. Matthew’s University School of Medicine, Grand Cayman Islands, BWI 4Department of Biochemistry, K.M.C.T. Medical College, Calicut University, Calicut, India
Received: 17 Feb 2011 Accepted: 26 Mar 2011
*Address correspondence and reprints request to: B.V. Murlimanju, Department of Anatomy, Kasturba Medical College, Manipal University, Mangalore, India. E-mail: murali.manju@manipal.edu |
|
| |
Abstract
Thanatophoric dysplasia is the lethal skeletal dysplasia characterized by marked underdevelopment of the skeleton and short-limb dwarfism. The child will be having a short neck, narrow thoracic cage and protuberant abdomen. Other anatomical features include a relatively enlarged head with frontal bossing, prominent eyes, hypertelorism and the depressed nasal bridge. The diagnosis is usually made with the ultrasonography in the second trimester. In this study we report a case of this rare entity with emphasis on its anatomical features, abnormalities and clinical profile with relevant review of literature.
Keywords: Thanatophoric dysplasia; FGFR3; Ultrasound; Fetus.
Introduction
Thanatophoric dysplasia (TD) is a type of neonatal lethal skeletal dysplasias.1 It is characterized by marked underdeveloped skeleton and short-limb dwarfism.1 The name TD is derived from the Greek word which means "death-bearing."2 TD is caused due to mutation of the fibroblast growth factor receptor 3 gene (FGFR3), which is located on the short arm of chromosome 4. The mutation results in the activation of FGFR3 tyrosine kinase independently of ligands such as fibroblast growth factor 8. This activation of FGFR3 results in decreased apoptosis and increased proliferation.1 It was reported that hypochondroplasia, achondroplasia and thanatophoric dysplasia are the different types of mutation in FGFR3 with hypochondroplasia being the mildest and TD, the most severe form.3 Since the clinical profile of this anomaly was rarely reported, we discuss the anatomical features, abnormalities and clinical profile of TD in the present report.
Case Report
In a Rural Hospital at Kerala State of India, a dead male fetus with many anatomical abnormalities was examined. The mother aged 25 yrs with second gravida and 22 weeks gestation was admitted to the hospital with abdominal pain. Routine ultrasonography revealed a single dead fetus in cephalic presentation with mild hydrocephalus and brachycephaly. The ultrasound also reported anterior narrowing of the skull, short ribs, narrow thorax, increased nuchal translucency, frontal bossing, short fetal limbs and protuberant abdomen. The other features observed include absence of corpus callosum, flattened vertebral bodies, narrow spinal canal. The ultrasound was reported as TD with cloverleaf skull deformity. Then the patient was advised termination of pregnancy. The fetus was examined macroscopically; the anatomical abnormalities were noted and were returned to the mother. On history taking, it was known that the couples had first degree consanguineous marriage and there was no past or family history of congenital abnormalities. On inspection, the fetus (Fig. 1) had large head, frontal bossing, short neck, narrow chest, protruded abdomen and short limbs. The trunk length was normal, but there was anterior narrowing of the skull. After discussing with the pediatrician and gynecologist, the fetus was diagnosed to have TD type 1. In the present report, the written informed consent was taken for the examination.
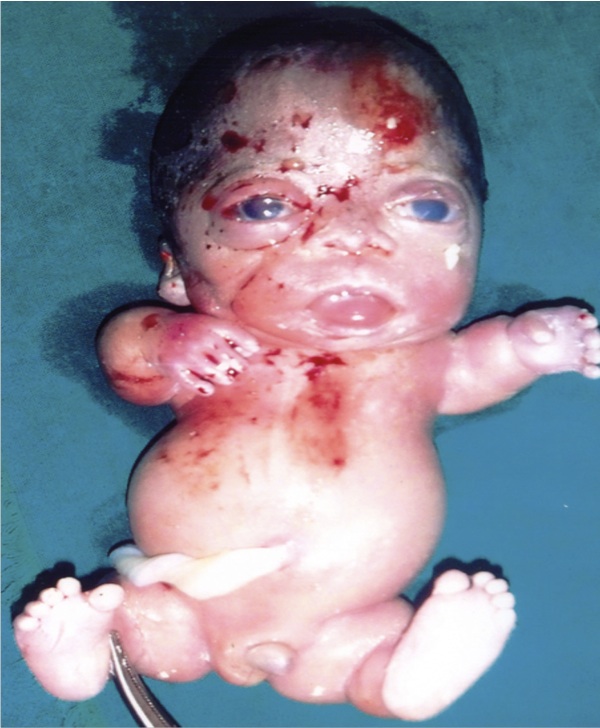
Figure 1: The Thanatophoric dysplasia in the fetus of 22 weeks of gestational age. The short neck, small thoracic cage, protuberant abdomen and short limbs were the typical features.
Discussion
Thanatophoric dysplasia or dwarfism (TD) is characterized by severe limb shortening (micromelia), bowing of limbs, narrow thorax and protuberant abdomen.4 Other features include polyhydramnios, large head, frontal bossing, cloverleaf skull, prominent eyes, hypertelorism, small pelvis and a depressed nasal bridge. It was observed that the abnormal temporal lobe development is a common associated feature and can be visualized as early as the second trimester on ultrasound.1 This entity has an estimated incidence of 1 in 20,000 to 50,000 births and is an autosomal dominant condition.5 There have been two types of TD observed. Type I TD is characterized by marked underdeveloped skeleton and short-curved long bones. The pelvic bones and vertebral columns are underdeveloped, cloverleaf skull may or may not be present.6 Whereas in type II TD, the long bones are not as short as in type I and are not bent. The fetuses with type II TD are reported to have cloverleaf skull which means a trilobed skull. The premature closures of coronal and lambdoid sutures are commonly seen with the cloverleaf skull.1
As the name thanatophoric or "death-bearing" suggests, the condition is frequently lethal in-utero or shortly after birth. The cause of death is respiratory failure that occurs shortly after birth. Respiratory insufficiency may be secondary to the narrow chest cavity and hypoplastic lungs, brain stem compression by the narrow foramen magnum or a combination of both.5 Surviving individuals can only live to early childhood and only with intensive medical support. The prolonged suffering is not limited to the neonate concerned but also to the parents as the surviving neonate is almost always ventilator-dependent and mentally deficient.5 Correct diagnosis allows proper genetic counseling and prognostication. It was reported that, with modern day obstetrics care, full term TD is less commonly seen. The diagnosis is usually suspected by antenatal ultrasound examination and confirmed by molecular analysis on amniocytes.5
The counseling part of management is very important in this disease, especially to the parents of the affected children. Without proper genetic counseling, most families having had a fetus with TD would be too worried to have further pregnancy.5 Since the majority of cases of TD occur sporadically, it is important to counsel that the recurrence risk is low for only one previously affected fetus and that the extended family members of the proband are not at increased risk.5 A general empiric recurrence risk for this entity was estimated as only 2%. To relieve the parental anxiety in such low risk couple, prenatal ultrasound examination may be offered in subsequent pregnancies to identify features suggestive of TD, such as macrocephaly, vertebral ossification defect, bowed femori, micromelia, and small thorax with protuberant abdomen. If indicated, amniocentesis may be offered and the diagnosis may be done by molecular analysis.5 It was reported that the prenatal diagnosis of TD has been well established with ultrasound examination usually in the second trimester.7 The 3D anatomy scan and molecular confirmation may be helpful in early diagnosis and genetic counseling of TD.8
Conclusion
In the present case, we observed that the fetus (Fig. 1) was having frontal bossing, large head, short neck, narrow chest, protuberant abdomen and short limbs. The anterior narrowing of the skull was present, but the trunk length was normal. The fetus was diagnosed to have TD type I. The detail regarding this anomaly was rarely reported in the literature. So we believe that this study has provided optimum knowledge on this subject and is important for the budding medical students, doctors especially the obstetricians and pediatricians. The report is also useful to the clinical anatomists, embryologists and morphologists.
Acknowledgements
The authors reported no conflict of interest and no funding was received on this work.
|
|
| |
1. Miller E, Blaser S, Shannon P, Widjaja E. Brain and bone abnormalities of thanatophoric dwarfism. AJR Am J Roentgenol 2009 Jan;192(1):48-51.
2. Orioli IM, Castilla EE, Barbosa-Neto JG. The birth prevalence rates for the skeletal dysplasias. J Med Genet 1986 Aug;23(4):328-332.
3. Cohen MM Jr. Achondroplasia, hypochondroplasia and thanatophoric dysplasia: clinically related skeletal dysplasias that are also related at the molecular level. Int J Oral Maxillofac Surg 1998 Dec;27(6):451-455.
4. Fink IJ, Filly RA, Callen PW, Fiske CC. Sonographic diagnosis of thanatophoric dwarfism in utero. J Ultrasound Med 1982 Oct;1(8):337-339.
5. Lam AC, Lam YY, Tong TM, et al. Thanatophoric dysplasia type 1 (TD1) without "telephone receivers". HK J Paediatr 2006;11:320-323.
6. Langer LO Jr, Yang SS, Hall JG, Sommer A, Kottamasu SR, Golabi M, et al. Thanatophoric dysplasia and cloverleaf skull. Am J Med Genet Suppl 1987;3:167-179.
7. Machado LE, Bonilla-Musoles F, Osborne NG. Thanatophoric dysplasia. Ultrasound Obstet Gynecol 2001 Jul;18(1):85-86.
8. Wong HS, Kidd A, Zuccollo J, Tuohy J, Strand L, Tait J, et al. A case of thanatophoric dysplasia: the early prenatal 2D and 3D sonographic findings and molecular confirmation of diagnosis. Fetal Diagn Ther 2008;24(1):71-73.
|
|