| |
Abstract
Objectives: The aim of this study is to investigate the effect of eicosapentaenoic acid combined with vitamin C in comparison with the pure form of eicosapentaenoic acid on the serum concentration of malondialdehyde, erythrocyte activity of superoxide dismutase, glutathione peroxidase, and the serum level of total antioxidant capacity in patients with type 2 diabetes.
Methods: Eighty one male diabetic patients, aged 33-63 years, were randomly assigned to one of 4 groups. The subjects consumed 500 mg/d pure eicosapentaenoic acid, 200 mg/d vitamin C, 500 mg eicosapentaenoic acid and 200 mg/d vitamin C or placebo depending on their groups. In fasting blood samples, superoxide dismutase and glutathione peroxidase activities were determined via the enzymatic method (Randox kit) and the serum total antioxidant capacity, malondialdehyde and vitamin C concentrations were estimated by colorimetric methods.
Results: Administration of pure eicosapentaenoic acid in diabetic patients increased superoxide dismutase by 4%, glutathione peroxidase 53%, total antioxidant capacity 36% and decreased malondialdehyde significantly by 25%. Prescription of eicosapentaenoic acid combined with vitamin C demonstrated a significant increment for superoxide dismutase activity by 3% and for glutathione peroxidase activity by 52% during the study, but no significant change was seen for total antioxidant capacity and malondialdehyde, respectively. There was a significant decrease in FBS and HbA1c following prescription of eicosapentaenoic acid with/without vitamin C along the study, although these changes were not significant between the study groups.
Conclusion: It is concluded that prescription of eicosapentaenoic acid in the pure form reduces oxidative stress in type 2 diabetic patients; albeit, it does not alleviate hyperglycemia. Combination of vitamin C and eicosapentaenoic acid does not improve antioxidant property of eicosapentaenoic acid.
Keywords: Diabetes; Eicosapentaenoic acid; Glutathione peroxidase; Superoxide dismutase; Total antioxidant capacity; Malondialdehyde.
Introduction
Diabetes mellitus, recognized by elevation of blood glucose concentration and lipid abnormalities, is accompanied by mitochondrial oxidative injuries.1 It has been reported that excessive blood glucose or FFAs trigger the development of reactive oxygen species (ROS) in muscle and adipose tissue.2-4 Oxidative injuries will possibly be the reason for cell dysfunction in diabetic patients.5,6 β-cells are also compromised from oxidative damage and β-cell apoptosis will be induced by the superoxide anions.5
Superoxide anions are enzymatically transformed to hydrogen peroxide by superoxide dismutase (SOD) within mitochondria and then hydrogen peroxide can be quickly removed by the mitochondrial enzyme glutathione peroxidase (GPx).7 Imbalance between the formation and break down of superoxide anions by mitochondrial enzymes in diabetes results in atherosclerosis and microvascular complications.8,9 Prescription of antioxidants such as vitamins C, E, lipoic acid and glutathione can reduce superoxide anions via modulating anti oxidant enzymes (SOD and GPx) activity.10-12
There are several reports that indicate n-3 fatty acid prescription increases antioxidant enzymes as well as reducing the risk of atherosclerosis in human studies,13,14 although some literature have shown an augmented lipid peroxidation susceptibility because of the high double bound content of n-3 fatty acids.15 These studies were based primarily on the results of the thiobarbituric acid assay (TBA) to asses malodialdehyde (MDA) level as a marker of highly unsaturated n-3 fatty acids oxidation.16 Ghiasvand et al. reported a significant increase in MDA after EPA supplementation in basketball players, where the concentration of MDA remained unchanged during the study in those treated with EPA combined with vitamin E.17
This current study, based on the incorporation of highly unsaturated n-3 fatty acids in lipid peroxidation,18 and the involvement of vitamin C in reducing the lipid peroxidation in diabetic patients,19 was prepared to investigate the effect of EPA combined with vitamin C in comparison with the pure form of EPA on the serum concentration of MDA, serum activity of SOD and GPx, as well as serum total antioxidant capacity (TAC) in patients with type 2 diabetes.
Methods
One hundred male subjects with type 2 diabetes, aged 33-63 years, were recruited from the public by media advertising. Entry criteria included: body mass index (in kg/m2) <35, no special diet, and no previous medical history of thyroid, liver, renal or chronic inflammatory diseases. None of the subjects were regularly taking lipid-lowering, beta-adrenergic antagonist or thiazide diuretic drugs. None of the subjects took fish oil supplements. Subjects were excluded if they had a recent history (within 3 months) of heart disease, angina, or major surgery; had a recent history (within 6 months) of myocardial infarction or stroke; had significant liver or renal disease (plasma creatinine >130 mol L-1), macroproteinuria, or symptomatic autonomic neuropathy; or regularly used nonsteroidal anti-inflammatory drugs. Eighty-one out of the total 100 subjects screened satisfied the entry criteria and completed the study. The study was approved by the Ethics Committee at Tehran University of Medical Sciences, and the reported investigations were carried out in accordance with the principles of the Declaration of Helsinki as revised in 2000. Informed consent was obtained from all individuals. The participants were instructed not to take any n-3 fatty acids and antioxidant supplements during and 8 weeks preceding the study.
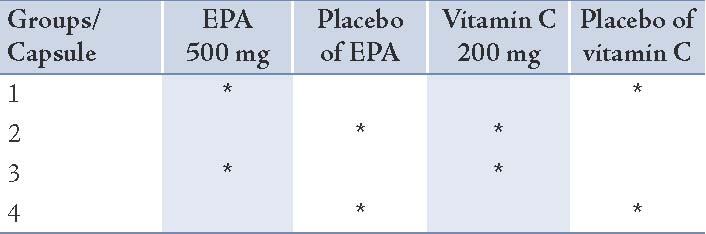
Baseline measurements were collected during a 3-week period, during which all subjects maintained their usual diet. They were randomly assigned to one of the 4 groups after stratification by age and body mass index. All participants received 2 soft gel capsules daily for 8 weeks: Group 1 took 500 mg EPA and the placebo of vitamin C; Group 2 received 200 mg vitamin C and the placebo of EPA; Group 3 was administered 500 mg EPA plus 200 mg vitamin C; and Group 4 was administered the corresponding placebo respectively. The details of the soft gels consumed in each group are given in Table 1. The subjects consumed EPA and/or vitamin C or placebo depending on their groups while continuing to consume their usual diet. EPA and the corresponding placebo soft gels were supplied from Minami Nutrition (Belgium). Vitamin C and the corresponding placebo soft gels were obtained from Darou Pakhsh Pharma Chem. Co., (Iran).
Subjects and investigators were blinded to the treatment. Subjects were instructed not to change their usual diets, level of physical activity, or other lifestyle factors throughout the intervention period. Before the baseline period, a dietitian gave written and verbal instructions to the subjects on how to keep accurate dietary records, including how to weigh or measure food. A 3-day dietary record (2 week days and 1 weekend day) and a lifestyle questionnaire including history of illness, medications and physical activity were completed at baseline and after the intervention period. Physical activities of participants were evaluated by a questionnaire based on the International Physical Activity Questionnaire, 2005 Epic-Norfolk. Each food and beverage was analyzed for content of energy and other nutrients using NUNTRITIONIST III software (version 7.0; N-Squared Computing, Salem, OR), which was designed for Iranian foods. Weight and height of subjects were measured by a digital scale and non-stretchable meter. Weight, changes in physical activity, medication, and any illness were recorded each week during baseline and at weeks 2, 4, 6 and at the end of the intervention.
For the biochemical measurements, fasting blood samples were taken at baseline and at the end of the 8 week study; all EDTA treated blood samples were promptly centrifuged. Plasma and erythrocytes were separated and erythrocytes were washed three times with 0.9% NaCl solution, and then hemolyzed with four volumes of cold distilled water. Analyses were carried out at the laboratory of the department of nutrition and biochemistry on the day of blood collection using Selectra 2 autoanalyzer (Vital Scientific, Spankeren, The Netherlands). Serum glucose was determined by the glucose analyzer (YSI, Yellow Springs, OH). Glycosylated hemoglobin (HbA1c) levels were measured by immuneturbidimetric immunoassay (Unimate; Roche Diagnostics, Indianapolis, IN) with a normal range of 4.5-6.1%. Serum triglycerides were measured by using a glycerol-3 phosphate oxidase phenol aminoantipyrine, with an automated Technicon Axon Analyzer (Bayer Diagnostics, Sydney, Australia) using an enzymatic method. Measurement of erythrocyte activity of SOD was done using xanthine oxidase enzyme and the reagent [2-(p-iodophenol)-3-(p-nitrophenyl)-5-phenyl tetrazolium chloride (INT)] via enzymatic method (Randox kit, Inc. USA).20 GPx activity of erythrocytes lysate was determined using hydrogen peroxide and it was expressed in micromoles of NADPH/gram of hemoglobin for erythrocytes (Randox kit, Inc. USA).21 Serum MDA concentrations were determined using the thiobarbituric acid method.22
Total plasma antioxidant capacity was determined by new generation colored 2,2'-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid radical cation method), (ABTS), (Randox kit, Inc. USA).23 The serum vitamin C concentration was determined in venous blood under nonfasting conditions by the 2,3-dinitrophenylhydrazine method with calorimetric analysis.24 The inter and intra-assay were less than 3.5% at all. Statistical analysis was performed with an IBM computer using the SPSS 15 statistical software package (SPSS Inc., Chicago, IL). The distribution of variables was studied using probability plots and the Shapiro-Wilk test. Baseline demographic and biochemical values between groups were compared using ANOVA. Bonferroni correction was used wherever there was a main effect. The differences between groups in ROS scavenging enzymes, MDA, TAC or serum concentration of fasting blood sugar, TG and vitamin C were analyzed by ANCOVA. Adjustments for age, BMI and baseline values were made. Paired t-test was used to compare variables within the groups. A value of p<0.05 was accepted as significant.
Results
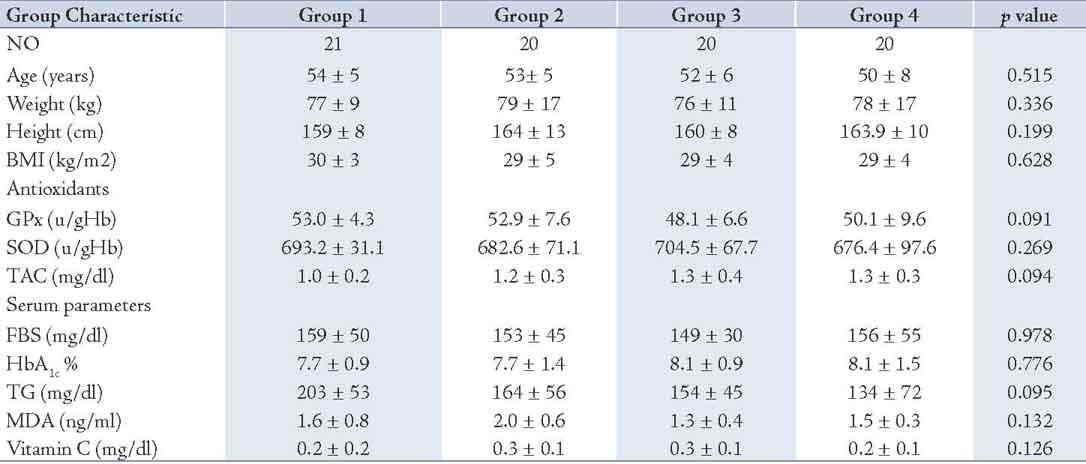
Eighty one out of the total 100 subjects screened satisfied the entry criteria and completed the study. The FBS and HbA1c levels in study subjects were 154.49 ± 45.69 and 7.89 ± 1.24, respectively, which showed all participants were under fairly good glycemic control. The characteristics of the patients confirmed that the groups were well matched for all entry criteria (Table 1), and there were no significant differences between the groups in either the type or the number of antihypertensive or oral hypoglycemic medications. The antihypertensive medications taken by the subjects were angiotensin-converting enzyme inhibitors (15%), calcium channel blockers (8%), angiotensin II receptor antagonists (10%), alpha-blockers (5%) and non thiazide diuretics (5%).
Table 1: The details of the soft gels consumed in each group.
There were no significant differences between the groups in total energy intake, macronutrient intake, body weight, or the number of cigarettes smoked at baseline or during the intervention (data not shown). Medication doses and physical activity were unchanged during the intervention in each group. As for the serum scavenging enzymes FBS, HbA1c and TG, the fasting triglyceride concentrations at baseline were <500 mg dL-1 in each of the groups. There were no significant differences between the groups at baseline in TG, FBS, HbA1c, SOD, GPx, MDA, vitamin C or TAC. (Table 2)
Table 2: Baseline demographic and biochemical characteristics of the patients (n= 81).
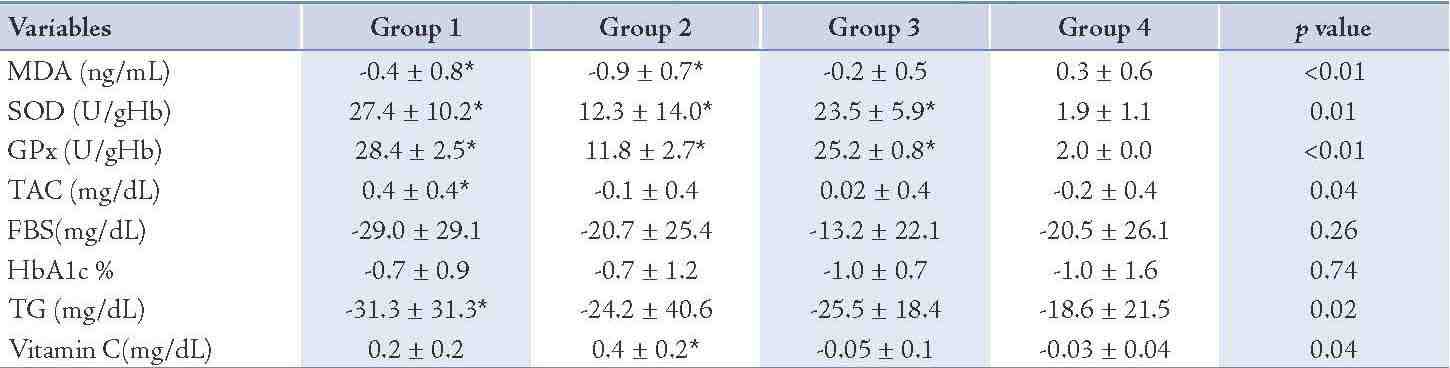
After 8 weeks administration of EPA dose of 500 mg/d, the concentration of MDA decreased significantly (25%; p=0.04) and the activity of the SOD and GPx as well as TAC increased (4%; p<0.01), (53%; p<0.01) and (36%; p<0.01), properly during intervention (Table 3). The changes in MDA, SOD, GPx and TAC were significantly different between group 1 and group 4, respectively (Table 4). The levels of TG, FBS and HbA1c decreased (15%; p<0.01), (18%; p<0.01), (9%; p<0.01) properly and vitamin C enhanced (70%; p<0.01) in group 1 (Table 3), whereas these changes in FBS, HbA1c and vitamin C concentration in group 1 in comparison to group 4 were not significant. (Table 4)
The vitamin C prescription at the dose of 200 mg/d significantly reduced the serum MDA levels (44%; p<0.01), the serum concentration of FBS (14%; p<0.01), HbA1c (9%; p=0.03) and the serum TG concentration (15%; p=0.02) and increased the erythrocyte SOD activity (2%; p<0.01), the erythrocyte GPx activity (22%; p<0.01) and the serum concentration of vitamin C (137%; p<0.01) during the study (Table 3). There was no significant change in the serum concentration of TAC after intervention (Table 3). The changes in SOD and GPx erythrocyte activity as well as serum vitamin C concentration were significantly different between groups 2 and 4 (Table 4). The total antioxidant capacity, TG, FBS and HbA1c did not show significant differences between group 2 and group 4. (Table 4)
The EPA supplementation of 500 mg/d plus 200 mg vitamin C showed a significant increment for SOD (3%; p<0.01), for GPx (52%; p<0.01) and a significant decrease of 9% (p=0.03) for FBS, 12% (p<0.01) for HbA1c and (17%; p<0.01) for TG during the study (Table 3). No significant changes were seen in the concentrations of MDA, vitamin C and TAC before and after the intervention in group 3 (Table 3). The changes in SOD and GPx were significantly different in group 3 compared to group 4 (Table 4), but there was no significant change in MDA, TAC, FBS, HbA1c, TG and vitamin C serum concentrations in group 3 compared to group 4. (Table 4)
Table 3: Changes in variables along among the study groups following intervention.
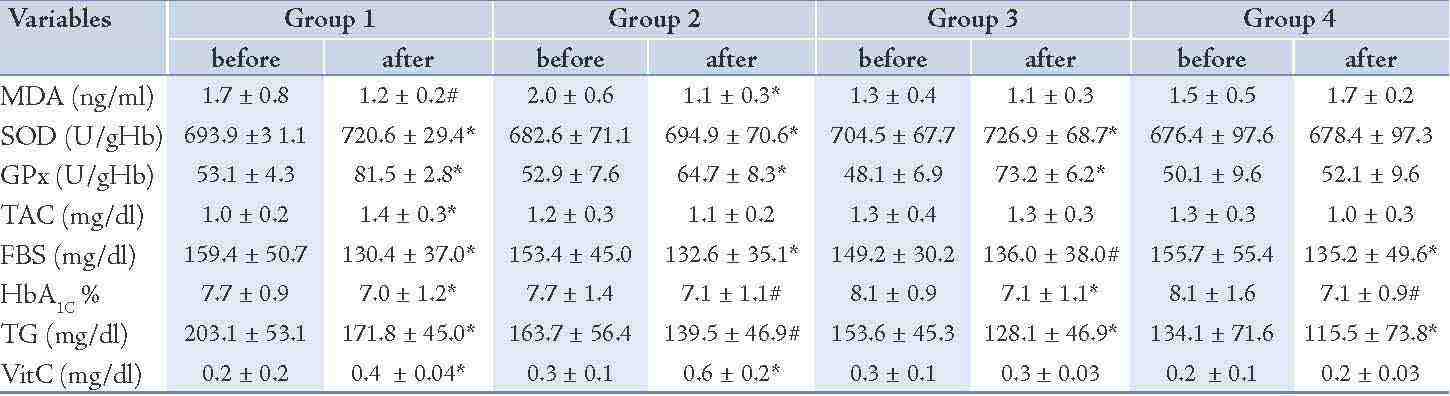
Table 4: Differences in variables at the end of the study following intervention between the groups.
Discussion
Glucotoxicity and lipotoxicity are culprit of oxidative stress in diabetic patients.5,6 In this study, EPA prescription in the pure form decreased the serum concentration of MDA in diabetic patients. In accordance with present findings, a significant reduction in MDA concentration has been reported by Kesavulu sequel n-3 fatty acids prescription in diabetic patients.25 The antioxidative properties of the pure EPA in this study could be attributed to significant reduction of TG concentrations following EPA prescription, which alone can play a major role in the prevention of lipid oxidation. Accordingly, it has previously been reported that a lipid-lowering agent, gemfibrozil, significantly reverses the excess lipid peroxidation in plasma and vascular tissue without significant changes in glucose metabolism and antioxidant enzymes in diabetic rats.26
Brude et al. in corroboration with current results reported significant reduction of MDA levels following EPA prescription in diabetic patients. They believed that the reduced oxidation rates following n-3 fatty acids consumption were due to tight packing of EPA and docosahexaenoic acid (DHA) in the lipid membrane complex which provides fewer double bonds for interaction with free radicals.27 Higgins and Brude advocated that the antioxidant property of n-3 fatty acids based on increased plasma and platelet concentrations of β-carotene as an antioxidant marker and reduction of MDA is observed only in long term studies (more than 6 weeks). They believed that adaptive compensatory mechanisms that are involved in decreased lipid peroxidation are activated only in long-term intervention in contrast to short term supplementation of n-3 fatty acids.27,28 Decreased inflammations and execution of free radical formation in white blood cells following n-3 fatty acid consumption has also been assumed by Trevor to explain the reduction of lipid peroxidation.29 The combination of vitamin C and EPA in this research showed no significant changes in the concentration of MDA during the study. In line with the result of this study, Sarbolouki et al. depicted an insignificant change in serum MDA level sequel to the prescription of EPA combined with other antioxidants in diabetic patients.30
In this research, the administration of EPA in its pure form as well as in conjunction with vitamin C increased the erythrocyte SOD activity. In accordance with the present study, Rahman et al. advocated an elevated level of SOD in mice after raising n-3 fatty acid serum concentration.12 Other findings indicate that EPA may reduce ROS production via upregulation of mitochondrial regulators of hydrogen peroxide generation.31 Another mechanism explained the enhancement of SOD following EPA prescription involves the findings of Rahman et al. which demonstrated a positive significant association between n-3 fatty acids intake and higher levels of sirtuin expression.13 It has been reported that sirtuin regulates the enzymatic properties of SOD in detoxifying mitochondrial superoxide.32
The GPx is another mitochondrial antioxidant enzyme that has been increased significantly along the study following prescription of EPA combined with vitamin C or without vitamin C. In corroboration with the present study, a significant elevated GPx activity has been shown by Kesavulu et al. in diabetic patients after n-3 fatty acids prescription.25 After 8 weeks of EPA prescription in the pure form not in combination with vitamin C, the total antioxidant activity was significantly augmented. The current results agree with the findings of García-Alonso et al. which implicate that the serum antioxidant status improved following n-3 fatty acids prescription, and revealed an increase in total antioxidant capacity.14 EPA participates in cell membrane phospholipids and intact lipoproteins such as low density lipoprotein (LDL). Membrane phospholipids are substrates for the production of second messengers resembling diacylglycerol, and it has been demonstrated that the fatty acid composition of such second messengers, which is formed by the precursor phospholipid, can manipulate their antioxidant properties.33
The data from the current study indicates that the fasting blood glucose did not change between the groups after intervention. There are controversies concerning the impact of n-3 fatty acids on blood sugar.34,35 Woodman and colleagues showed augmentation of fasting blood sugar after supplementation of 4000 mg EPA and 4000 mg DHA.35 It seems higher doses of EPA prescription have been associated with increments of FBS in animal and human researches.35 Furthermore, these discrepancies could be explained by genetic variations in the population of the studies. Schafer and his group showed genetic variants of the fatty acid desaturase 1/2 gene cluster that differently convert n-3 and n-6 PUFA catalyzed by the Δ5 and Δ6 desaturase.36
The relationship between type 2 diabetes and higher triglyceride levels as well as the hypotriglyceridemic effect of n-3 fatty acids are well established and could be found elsewhere.37,38 In this study, a similar result based on the reduced TG concentration following EPA prescription was found. A mechanism to explain the hypotriglyceridemic effects of n-3 fatty acids in humans has not been clarified. Recently, a working model was developed at the gene transcriptional level, which involves four metabolic nuclear receptors.39 These include liver X receptor, hepatocyte nuclear factor-4 alpha (HNF-4 alpha), farnesol X receptor, and peroxisome proliferators-activated receptor (PPARs). Each of these receptors is regulated by sterol receptor element binding protein-1c (SREBP-1c), a hepatic gene transcription factor that stimulates synthesis of the lipogenic enzymes involved in this pathway.40 N-3 fatty acids elicit hypotriglyceridemic effects by coordinately suppressing hepatic lipogenesis through reducing levels of SREBP-1c, upregulating fatty oxidation in the liver and skeletal muscle through PPAR activation, and enhancing flux of glucose to glycogen through down regulation of HNF-4 alpha. The net result is the repartitioning of metabolic fuel from triglyceride storage toward oxidation, thereby reducing the substrate available for Very-Low-Density Lipoprotein (VLDL) synthesis.39 Interestingly, if the consequences of reduced TG concentration decrease the oxidation of fatty acids, intramitochondrial NADH/NAD+ ratio diminishes and the formation of ROS in different cells lessen.2-4
In this research, administration of vitamin C showed a significant increase in SOD and GPx erythrocyte activity. Vitamin C is able to modulate immune responses in several ways, for instance, by modulating leukocyte function and lymphocyte proliferation.41 There are several reports in keeping with this study demonstrating a significant enhancement in SOD and GPx activity following vitamin C supplementation.42 Bernardo et al. have recently reported that administration of vitamin C in small-bowel mucosal biopsy organ culture system prevents free radical cytotoxicity and enhances scavenging enzymes such as superoxide dismutase, glutathione peroxidase and catalase.43 The second line of defense against oxidant-induced cellular injury is provided by low molecular weight scavengers such as thiols, especially reduced glutathione, alpha-tocopherol (vitamin E), vitamin C, carotenoids, methionine and taurine.44 Frei et al. showed that ascorbic acid is more effective than protein thiols, bilirubin, urate or tocopherol, as an antioxidant in the plasma with an in vitro system.45 The results of this study also showed more reduction in the serum MDA concentration after EPA supplementation compared to vitamin C. (Table 4)
The findings in the current study showed that a combination of vitamin C and EPA did not improve antioxidant properties of EPA revealed by inert impact on serum MDA and TAC levels. It should be mentioned that vitamin C has pro-oxidative and antioxidative properties. Podmore et al. described the potential pro-oxidant effects of daily supplementation with 500 mg of vitamin C on DNA base oxidation in vivo.46 Rahman et al. also showed that iron and vitamin C co-supplementation increases oxidative damage to DNA in healthy volunteers.47 Based on this research, it is hypothesized that the pro-oxidative property of vitamin C may increase oxidation of highly double bound n-3 fatty acids and also eliminate the antioxidative influence of EPA recognized by the reduction of MDA.
It is worth mentioning that serum EPA concentration was not determined in the four groups at the beginning and throughout the study. As a limitation of this study, it should be taken into consideration that measuring EPA serum concentration at the beginning and during the study would make this study more valid.
Conclusion
Overall, the prescription of EPA at the dose of 500 mg/day did not improve hyperglycemia but it can reduce oxidative stress in type 2 diabetic patients by activating antioxidant enzymes (SOD, GPx) and increasing the total antioxidant capacity. Vitamin C in accordance with EPA has shown antioxidative characteristics in type 2 diabetic patients. A combination of vitamin C and EPA did not improve the antioxidant property of EPA.
Acknowledgements
This study was supported in part by the Institute of Public Health Research, Tehran University of Medical Sciences, Tehran, Iran. (DP/8703277330,14/9/2010). We wish to thank Minami Nutrition (Belgium) company for preparing the EPA soft gels. Authors reported no conflict of interest for this work.
References
1. Green K, Brand MD, Murphy MP. Prevention of mitochondrial oxidative damage as a therapeutic strategy in diabetes. Diabetes 2004 Feb;53(1)(Suppl 1):S110-S118.
2. Talior I, Yarkoni M, Bashan N, Eldar-Finkelman H. Increased glucose uptake promotes oxidative stress and PKC-δ activation in adipocytes of obese, insulin-resistant mice. Am J Physiol Endocrinol Metab 2003 Aug;285(2):E295-E302.
3. Brownlee M. The pathobiology of diabetic complications: a unifying mechanism. Diabetes 2005 Jun;54(6):1615-1625.
4. Haber EP, Procópio J, Carvalho CR, Carpinelli AR, Newsholme P, Curi R. New insights into fatty acid modulation of pancreatic β-cell function. Int Rev Cytol 2006;248:1-41.
5. Kahn SE. The relative contributions of insulin resistance and β-cell dysfunction to the pathophysiology of Type 2 diabetes. Diabetologia 2003 Jan;46(1):3-19.
6. Kajimoto Y, Kaneto H. Role of oxidative stress in pancreatic β-cell dysfunction. Ann N Y Acad Sci 2004 Apr;1011:168-176.
7. Turrens JF. Mitochondrial formation of reactive oxygen species. J Physiol 2003 Oct;552(Pt 2):335-344.
8. Velázquez E, Winocour PH, Kesteven P, Alberti KG, Laker MF. Relation of lipid peroxides to macrovascular disease in type 2 diabetes. Diabet Med 1991 Oct;8(8):752-758.
9. Jennings PE, McLaren M, Scott NA, Saniabadi AR, Belch JJ. The relationship of oxidative stress to thrombotic tendency in type 1 diabetic patients with retinopathy. Diabet Med 1991 Nov;8(9):860-865.
10. Major TC, Overhiser RW, Panek RL. Evidence for NO involvement in regulating vascular reactivity in balloon-injured rat carotid artery. Am J Physiol 1995 Sep;269(3 Pt 2):H988-H996.
11. Jones W, Li X, Qu ZC, Perriott L, Whitesell RR, May JM. Uptake, recycling, and antioxidant actions of alpha-lipoic acid in endothelial cells. Free Radic Biol Med 2002 Jul;33(1):83-93.
12. Freedman JE, Li L, Sauter R, Keaney JF JR. alpha-Tocopherol and protein kinase C inhibition enhance platelet-derived nitric oxide release. FASEB J 2000 Dec;14(15):2377-2379.
13. Rahman M, Halade GV, Bhattacharya A, Fernandes G. The fat-1 transgene in mice increases antioxidant potential, reduces pro-inflammatory cytokine levels, and enhances PPAR-γ and SIRT-1 expression on a calorie restricted diet. Oxid Med Cell Longev 2009 Nov-Dec;2(5):307-316.
14. García-Alonso FJ, Jorge-Vidal V, Ros G, Periago MJ. Effect of consumption of tomato juice enriched with n-3 polyunsaturated fatty acids on the lipid profile, antioxidant biomarker status, and cardiovascular disease risk in healthy women. Eur J Nutr 2012 Jun;51(4):415-424.
15. Oostenbrug GS, Mensink RP, Hardeman MR, De Vries T, Brouns F, Hornstra G. Exercise performance, red blood cell deformability, and lipid peroxidation: effects of fish oil and vitamin E. J Appl Physiol (1985) 1997 Sep;83(3):746-752.
16. Meydani M, Natiello F, Goldin B, Free N, Woods M, Schaefer E, et al. Effect of long-term fish oil supplementation on vitamin E status and lipid peroxidation in women. J Nutr 1991 Apr;121(4):484-491.
17. Ghiasvand R, Djalali M, Djazayery SA, Keshavarz SA, Hosseini M, Askari GH, et al. Effect of eicosapentaenoic Acid (EPA) and vitamin e on the blood levels of inflammatory markers, antioxidant enzymes, and lipid peroxidation in Iranian basketball players. Iran J Public Health 2010;39(1):15-21.
18. Rathod NR, Raghuveer I, Chitme HR, Chandra R. Free Radical Scavenging Activity of Calotropis gigantea on Streptozotocin-Induced Diabetic Rats. Indian J Pharm Sci 2009 Nov;71(6):615-621.
19. Lautt WW, Ming Z, Legare DJ. Attenuation of age- and sucrose-induced insulin resistance and syndrome X by a synergistic antioxidant cocktail: the AMIS syndrome and HISS hypothesis. Can J Physiol Pharmacol 2010 Mar;88(3):313-323.
20. Fridovich I. Superoxide radical and superoxide dismutases. Annu Rev Biochem 1995;64:97-112.
21. Paglia DE, Valentine WN. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 1967 Jul;70(1):158-169.
22. Richard MJ, Portal B, Meo J, Coudray C, Hadjian A, Favier A. Malondialdehyde kit evaluated for determining plasma and lipoprotein fractions that react with thiobarbituric acid. Clin Chem 1992 May;38(5):704-709.
23. Erel O. A novel automated direct measurement method for total antioxidant capacity using a new generation, more stable ABTS radical cation. Clin Biochem 2004 Apr;37(4):277-285.
24. Lowry OH, Lopez JA, Bessey OA. The determination of ascorbic acid in small amounts of blood serum. J Biol Chem 1945;160:609-615.
25. Kesavulu MM, Kameswararao B, Apparao Ch, Kumar EG, Harinarayan CV. Effect of omega-3 fatty acids on lipid peroxidation and antioxidant enzyme status in type 2 diabetic patients. Diabetes Metab 2002 Feb;28(1):20-26.
26. Ozansoy G, Akin B, Aktan F, Karasu C. Short-term gemfibrozil treatment reverses lipid profile and peroxidation but does not alter blood glucose and tissue antioxidant enzymes in chronically diabetic rats. Mol Cell Biochem 2001 Jan;216(1-2):59-63.
27. Brude IR, Drevon CA, Hjermann I, Seljeflot I, Lund-Katz S, Saarem K, et al. Peroxidation of LDL from combined-hyperlipidemic male smokers supplied with omega-3 fatty acids and antioxidants. Arterioscler Thromb Vasc Biol 1997 Nov;17(11):2576-2588.
28. Higgins S, Carroll YL, McCarthy SN, Corridan BM, Roche HM, Wallace JM, et al. Susceptibility of LDL to oxidative modification in healthy volunteers supplemented with low doses of n-3 polyunsaturated fatty acids. Br J Nutr 2001 Jan;85(1):23-31.
29. Mori TA, Beilin LJ. Long-chain omega 3 fatty acids, blood lipids and cardiovascular risk reduction. Curr Opin Lipidol 2001 Feb;12(1):11-17.
30. Sarbolouki Sh, Djalali M, Dorosty A, Djazayery S, Eshraghian M, Ebadi S, et al. Effects of EPA and Vitamin E on Serum Enzymatic Antioxidants and Peroxidation Indices in Patients with Type II Diabetes Mellitus. Iran J Public Health 2010;39(3):82-91.
31. Takahashi M, Tsuboyama-Kasaoka N, Nakatani T, Ishii M, Tsutsumi S, Aburatani H, et al. Fish oil feeding alters liver gene expressions to defend against PPARalpha activation and ROS production. Am J Physiol Gastrointest Liver Physiol 2002 Feb;282(2):G338-G348.
32. Tao R, Coleman MC, Pennington JD, Ozden O, Park SH, et al. Sirt3-Mediated Deacetylation of Evolutionarily Conserved Lysine 122 Regulates MnSOD Activity in Response to Stress. Mole. cell. 2010 Dec 22; 40(6):893-904.
33. Miles EA, Calder PC. Modulation of immune function by dietary fatty acids. Proc Nutr Soc 1998 May;57(2):277-292.
34. Dunstan DW, Mori TA, Puddey IB, Beilin LJ, Burke V, Morton AR, et al. The independent and combined effects of aerobic exercise and dietary fish intake on serum lipids and glycemic control in NIDDM. A randomized controlled study. Diabetes Care 1997 Jun;20(6):913-921.
35. Woodman RJ, Mori TA, Burke V, Puddey IB, Watts GF, Beilin LJ. Effects of purified eicosapentaenoic and docosahexaenoic acids on glycemic control, blood pressure, and serum lipids in type 2 diabetic patients with treated hypertension. Am J Clin Nutr 2002 Nov;76(5):1007-1015.
36. Schaeffer L, Gohlke H, Müller M, Heid IM, Palmer LJ, Kompauer I, et al. Common genetic variants of the FADS1 FADS2 gene cluster and their reconstructed haplotypes are associated with the fatty acid composition in phospholipids. Hum Mol Genet 2006 Jun;15(11):1745-1756.
37. Al-Mukhtar SB, Fadhil NN, Hanna BE. General and gender characteristics of type 2 diabetes mellitus among the younger and older age groups. Oman Med J 2012 Sep;27(5):375-382.
38. He K, Liu K, Daviglus ML, Mayer-Davis E, Jenny NS, Jiang R, et al. Intakes of long-chain n-3 polyunsaturated fatty acids and fish in relation to measurements of subclinical atherosclerosis. Am J Clin Nutr 2008 Oct;88(4):1111-1118.
39. Davidson MH. Mechanisms for the hypotriglyceridemic effect of marine omega-3 fatty acids. Am J Cardiol 2006 Aug;98(4A):27i-33i.
40. Pégorier JP, Le May C, Girard J. Control of gene expression by fatty acids. J Nutr 2004 Sep;134(9):2444S-2449S.
41. Goetzl EJ, Wasserman SI, Gigli I, Austen KF. Enhancement of random migration and chemotactic response of human leukocytes by ascorbic acid. J Clin Invest 1974 Mar;53(3):813-818.
42. Hermsdorff HH, Barbosa KB, Volp AC, Puchau B, Bressan J, Zulet MÁ, et al. Vitamin C and fibre consumption from fruits and vegetables improves oxidative stress markers in healthy young adults. Br J Nutr 2012 Apr;107(8):1119-1127.
43. Bernardo D, Martínez-Abad B, Vallejo-Diez S, Montalvillo E, Benito V, Anta B, et al. Ascorbate-dependent decrease of the mucosal immune inflammatory response to gliadin in coeliac disease patients. Allergol Immunopathol (Madr) 2012 Jan-Feb;40(1):3-8.
44. Anonymous. Expanding knowledge of ascorbic acid metabolism. Nutr Rev 1989 Nov;47(11):360-361.
45. Frei B, England L, Ames BN. Ascorbate is an outstanding antioxidant in human blood plasma. Proc Natl Acad Sci U S A 1989 Aug;86(16):6377-6381.
46. Podmore ID, Griffiths HR, Herbert KE, Mistry N, Mistry P, Lunec J. Vitamin C exhibits pro-oxidant properties. Nature 1998 Apr;392(6676):559.
47. Rehman A, Collis CS, Yang M, Kelly M, Diplock AT, Halliwell B, et al. The effects of iron and vitamin C co-supplementation on oxidative damage to DNA in healthy volunteers. Biochem Biophys Res Commun 1998 May;246(1):293-298.
|