|
Abstract
The clinical presentation of coarctation of aorta (CoA) is well known; however, it is the most common congenital heart disease in the newborn period to be missed, with significant mortality and morbidity associated with missing the diagnosis. We report a 20-day-old newborn boy who presented with congestive heart failure and weak femoral pulses. Chest X-ray (CXR) showed cardiomegaly and pulmonary edema and electrocardiography (ECG) showed extreme right axis deviation and absent left ventricular forces in the left precordial leads. Based on these, clinical suspicion of coarctation of aorta was made and confirmed by echocardiography. After initial stabilization with prostaglandin E2 infusion, child underwent urgent coarctation of aorta repair with uneventful post-operative course. High index of suspicion for coarctation of aorta in a newborn with such presentation is required and urgent referral to Pediatric Cardiology and cardiac surgery center is mandatory to reduce the morbidity and mortality associated with missing the diagnosis.
Keywords: Coarctation of the aorta; Congenital heart disease.
Introduction
Coarctation of aorta (CoA) constitutes around 7% of all congenital heart diseases.1 The clinical presentation in the newborn period is classical and well described as in this case; however, several studies documented that it is the most common missed congenital heart disease in the newborn period.2-4 Missed diagnosis of CoA in the newborn period is associated with high morbidity and mortality,4,5 and prompt diagnosis and management is mandatory.
Case Report
A 20-day-old term baby with birth weight of 3.5 kg and uneventful prenatal and immediate postnatal history presented with history of interrupted feeding and fast breathing of 3 days’ duration. There was no history of fever or cough. He presented initially to a local hospital where he was treated as pneumonia, but the family presented to our hospital emergency department for a second opinion as there was no improvement in the child’s condition.
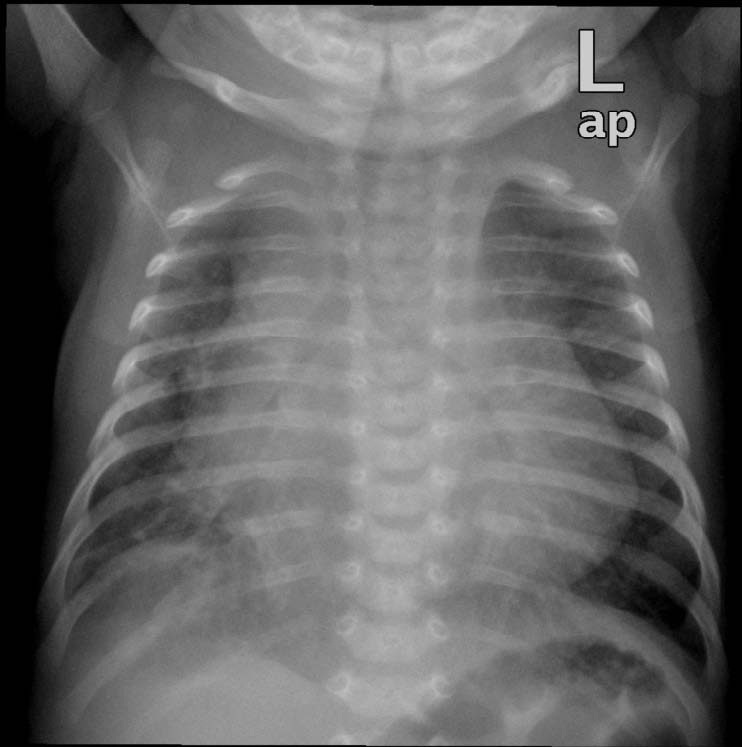
Figure 1: Anterior posterior chest x ray showing cardiomegaly with pulmonary edema.
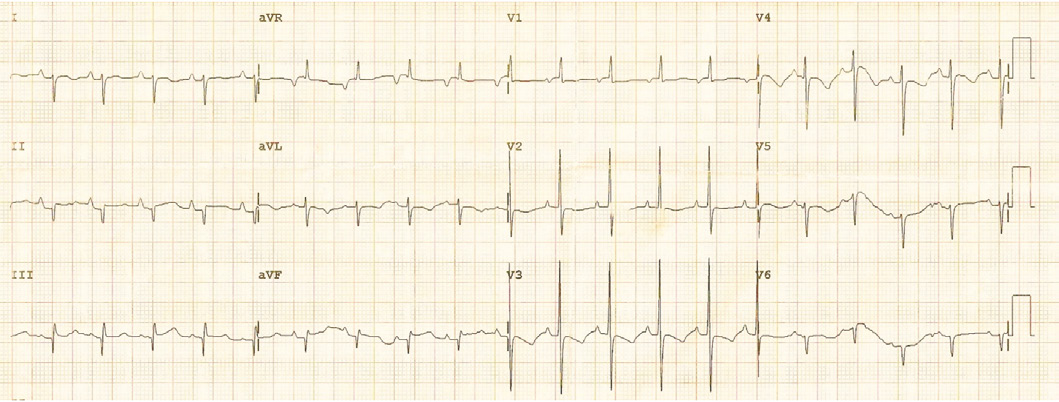
Figure 2: The 12-lead electrocardiogram showing marked right axis deviation with significant reduction in left ventricular forces in the lateral chest leads.
Physical examination showed no dysmorphic features with a weight of 3.5 kg. He was found to have mild respiratory distress with respiratory rate of 65 breaths/min, heart rate of 165 pulse/min, oxygen saturation in room air was 99% from right hand and 97% in left foot, temperature of 37°C and blood pressure in the right arm of 92/70, left arm of 90/67, right lower limb 72/68 and left lower limb of 72/64. Systemic examination revealed subcostal recessions with bilateral fine crepitations and hepatomegaly of 3 cm. Cardiac examination revealed normal heart sounds with no murmur and bilateral weak femoral pulses. Chest X-ray (CXR) showed cardiomegaly with cardio-thoracic ratio of 67% with evidence of pulmonary edema, (Fig. 1). Electrocardiography (ECG) showed sinus rhythm with features of extreme right axis deviation with absent left ventricular forces in leads V4 to V6, (Fig. 2). Echocardiography, (Fig. 3), confirmed the clinical suspicion with finding of severe juxtaductal coarctation of the aorta with peak gradient of 50 mmHg with severe dilation of the right atrium and right ventricle and moderate tricuspid valve regurgitation with peak gradient of 50 mmHg. There was no patent ductus arteriosus (PDA) flow seen. His ventricular systolic function was moderately reduced. He was started on prostaglandin infusion immediately, in addition to starting on preload reduction measures with furosemide and spironolactone and taken urgently for coarctation repair where he underwent resection of the coarctation and end-to-end anastomosis of the arch with PDA ligation. He was extubated on the 2nd postoperative day and discharged home on the 7th postoperative day with followup in outpatient clinic.
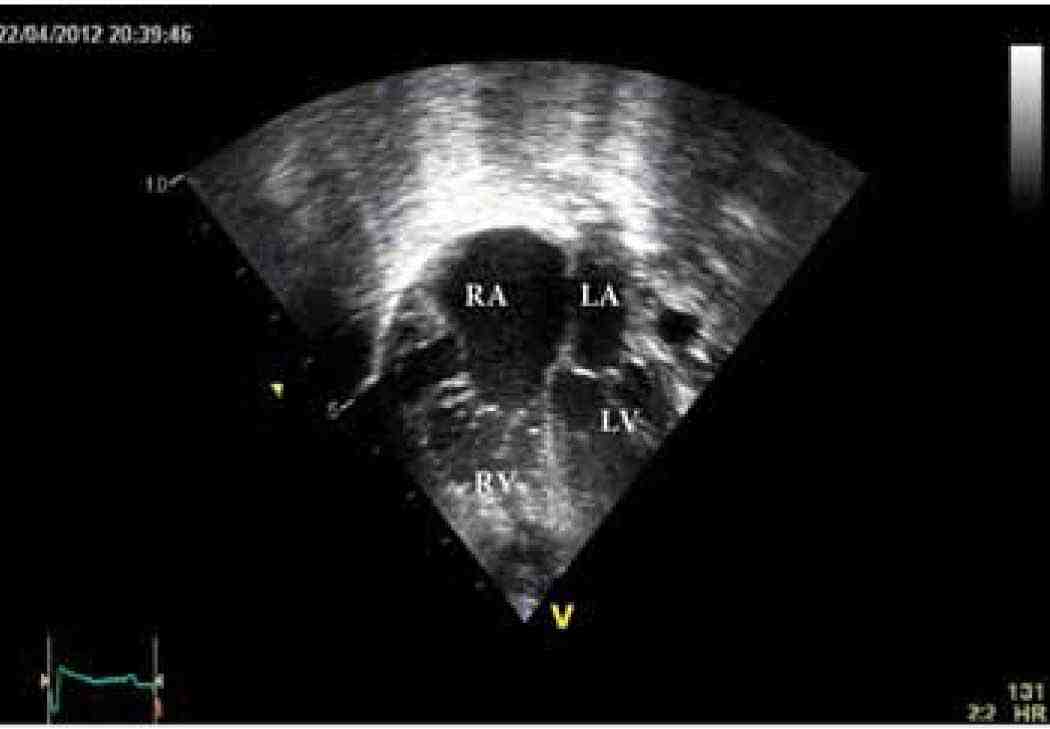
Figure 3a: Echocardiographic apical four-chamber view showing dilated right atrium (RA), dilated hypertrophied right ventricle (RV), left atrium (LA), and left ventricle (LV).
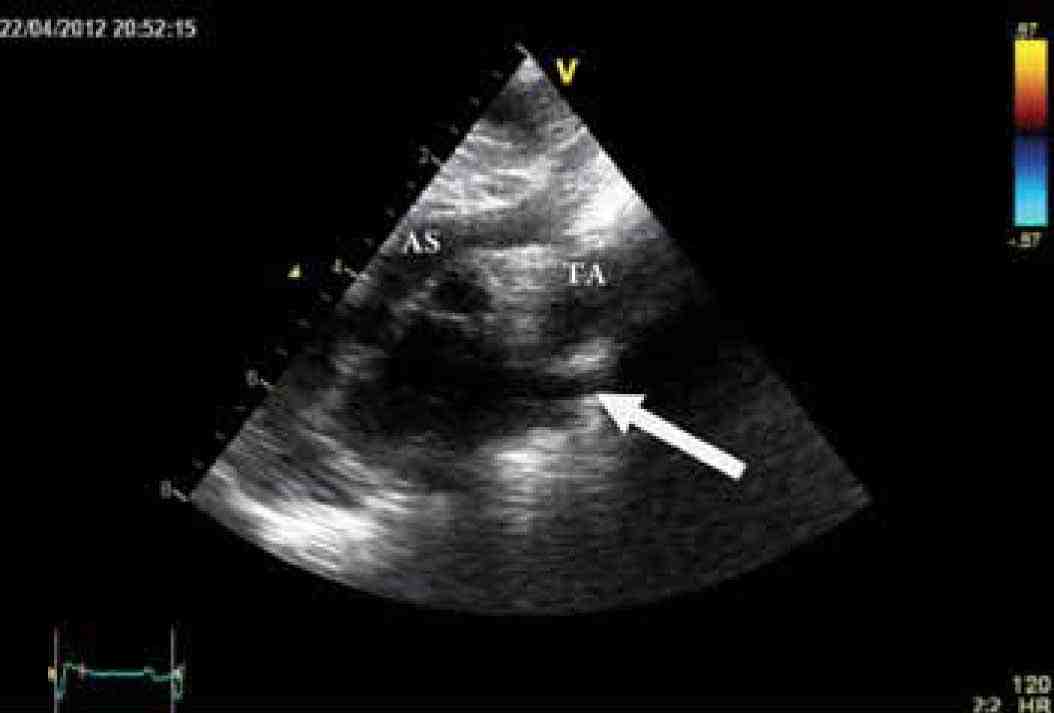
Figure 3b: Echocardiographic high parasternal view showing severe coarctation of aorta (arrow). Ascending aorta (AS), Transverse arch (TA).
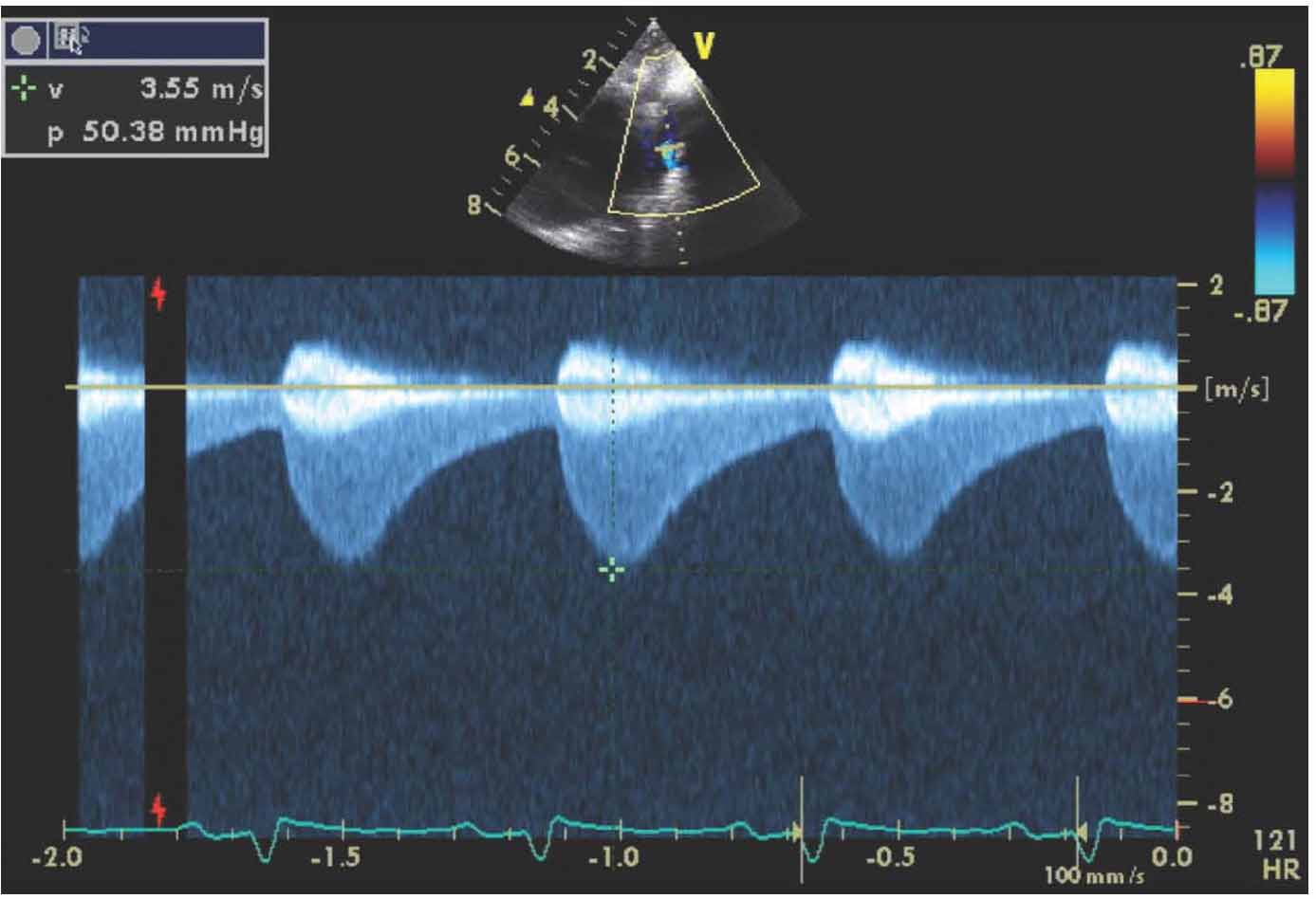
Figure 3c: Continuous-wave Doppler across aortic coarctation with prominent "diastolic tail," with peak systolic velocity of 50 mmHg.
Discussion
Most patients with coarctation of aorta appear well in the first 48 hours of life with normal newborn examination. Symptoms develop after discharge from newborn nursery. The extent of ductus arteriosus patency, and the rapidity of closure in addition to pulmonary vascular resistance will determine the timing and severity of presentation. This child's clinical presentation is classic for coarctation of aorta as the ductus arteriosus usually closes anatomically between 2 to 3 weeks of life,6 hence, pediatrician and general practitioners should be aware about the possibility of coarctation of aorta in a newborn with respiratory distress around this period of time.
Classically described, the narrowing could be proximal (preductal) to the ductus arteriosus and the blood flow in aorta distal to the narrowing is dependent on the ductus arteriosus and this is the most common type in neonates, hence, closure of the duct will result in early severe neonatal presentation. In post-ductal COA, the narrowing is distal to insertion of the ductus arteriosus and this is the common type in adult patients. Weak or absent femoral pulses are present in 92% of patients.3 Pulse oximetry measurement at pre- and post-ductal sites should be monitored. In case of critical coarctation of aorta, the preductal saturation is higher than the post-ductal saturation and differential cyanosis exist.7 The four limb blood pressures is an important examination tool and can identify discrepancy between upper and lower limb systolic blood pressure; however, this test lack specificity and discrepancy up to 20 mmHg could be normal which raise the importance of checking femoral pulses in all newborns.8 The electrocardiograph will show features of right ventricular hypertrophy although this could be normal in newborn period. The finding of extreme right axis deviation of 180 degrees or more or an axis in the right upper quadrant should raise the suspicion as these values are above the age appropriate reference range.9 During fetal life, the right ventricle is the systemic ventricle as the oxygenation is taking place in placenta. The right ventricle handles more blood when there is left side obstruction and compensatory hypertrophy will take place and this will result in features of right ventricle hypertrophy in ECG.
There is a long list of differential diagnoses for such presentation as left side obstructive lesions in the newborn period like for example interrupted aortic arch and hypoplastic left heart syndrome. The initial steps of management are hemodynamic stabilization with prostaglandin E2 infusion to open the ductus arteriosus which can be accomplished in 80% of patients up to age of 28 days. After starting the PGE2 infusion, patient should be monitored in critical care unit and periodic check for quality of femoral pulses and four limbs BP to check for response. Due to increase in plasma renin activity secondary to renal hypoperfusion, volume expansion should be treated by reducing preload with the use of diuretics. These measures should correct the metabolic acidosis which occur secondary to congestive heart failure. Postoperatively, such patients require a lifelong follow-up for any complications that might occur for example restenosis, aneurysm formation and development of systemic hypertension.10
Conclusion
High index of suspicion for coarctation of aorta in any newborn with signs of respiratory distress should be maintained and early detection can result in reduction in the morbidity and mortality associated with a late diagnosis.
Acknowledgements
The authors reported no conflict of interest and no funding was received for this work.
References
1. Kenny D, Hijazi ZM. Coarctation of the aorta: from fetal life to adulthood. Cardiol J 2011;18(5):487-495.
2. Chang RK, Gurvitz M, Rodriguez S. Missed diagnosis of critical congenital heart disease. Arch Pediatr Adolesc Med 2008 Oct;162(10):969-974.
3. Ing FF, Starc TJ, Griffiths SP, Gersony WM. Early diagnosis of coarctation of the aorta in children: a continuing dilemma. Pediatrics 1996 Sep;98(3 Pt 1):378-382.
4. Ward KE, Pryor RW, Matson JR, Razook JD, Thompson WM, Elkins RC. Delayed detection of coarctation in infancy: implications for timing of newborn follow-up. Pediatrics 1990 Dec;86(6):972-976.
5. Kishan J, Elzouki AY, Mir NA. Coarctation of the aorta in the newborn: a clinical study. Ann Trop Paediatr 1984 Dec;4(4):225-228.
6. Schneider DJ. The patent ductus arteriosus in term infants, children, and adults. Semin Perinatol 2012 Apr;36(2):146-153.
7. Mahle WT, Martin GR, Beekman RH III, Morrow WR; Section on Cardiology and Cardiac Surgery Executive Committee. Endorsement of Health and Human Services recommendation for pulse oximetry screening for critical congenital heart disease. Pediatrics 2012 Jan;129(1):190-192.
8. Crossland DS, Furness JC, Abu-Harb M, Sadagopan SN, Wren C. Variability of four limb blood pressure in normal neonates. Arch Dis Child Fetal Neonatal Ed 2004 Jul;89(4):F325-F327.
9. Rijnbeek PR, Witsenburg M, Schrama E, Hess J, Kors JA. New normal limits for the paediatric electrocardiogram. Eur Heart J 2001 Apr;22(8):702-711.
10. de Bono JP, Freeman LJ. Long term follow up of patients with repaired aortic coarctations. Heart 2005 Apr;91(4):537-538.
|