Large Brunner’s Gland Hamartoma: A Case Report
Uday Gokhale, G. Rajasekharan Pillai
ABSTRACT
Brunner’s gland hamartoma is an extremely uncommon benign tumor of the duodenum. Most of the lesions are small, asymptomatic and are detected incidentally. It may rarely attain a large size and produces a variety of clinical manifestations. Imaging modalities and endoscopy may locate the lesion, but definitive diagnosis requires histopathological examination. We report a case of a 58-year-old Omani female who presented with obstruction due to a large Brunner’s gland hamartoma.
From the Department Pathology, Sultan Qaboos Hospital, Salalah, Dhofar, Sultanate of Oman.
Received: 07 Aug 08
Accepted: 20 Oct 08
Address correspondence and reprint requests to: Dr. Uday A. Gokhale, Specialist, Department of Pathology, Sulatan Qaboos Hospital, Salalah, Dhofar, Sultanate of Oman.
E-mail: uday_gokhale@rediffmail.com
INTRODUCTION
Benign small bowel neoplasms are very rare. Brunner’s gland hamartoma, also known as Brunner’s gland adenoma or brunneroma, comprises a small portion of these benign tumors.1 It is a benign, polypoid proliferation of Brunner’s glands.2 Usually asymptomatic and discovered incidentally, these lesions may manifest occasionally as a cause of duodenal obstruction or upper gastrointestinal hemorrhage and require surgical excision.3
CASE REPORT
A 58-year-old Omani lady was admitted to the surgical ward with a two-month history of epigastric pain, abdominal discomfort, nausea and frequent bouts of vomiting. The patient had sought consultation about a month before, but did not respond to antacids and H2 blockers. The Complete Blood Count (CBC) and biochemical investigations were within normal limits. Computed Tomography (CT) of abdomen with contrast and barium meal study revealed a large polypoidal mass arising from the duodenal bulb which was further confirmed by endoscopy.
As the patient was symptomatic, she was subjected to laparoscopic removal of the polyp. Laparoscopic duodenotomy revealed a polyp with the pedicle attached to the first part of the duodenum and reaching up to the third part. It was excised and sent for histopathological examination.
PATHOLOGICAL FINDINGS
The resected specimen showed a light brown colored, firm, pedunculated, polypoid mass measuring 4.2 x 2.6 x 1.4 cm. The surface was smooth without any erosions or ulcer. The cut surface was firm, yellowish and lobulated with a few slit-like spaces.
On microscopic examination, the tumor was covered by small intestinal mucosa and composed of lobules of proliferated Brunner’s glands with ducts separated by irregular bands of fibromuscular stroma. (Figures 1a and 1b) There was mild infiltration by lymphocytes with a few lymphoid follicles. Some of the ducts were cystically dilated. (Figure 2) Paneth cell change was noted in some of the glands. The covering mucosa was flattened and showed congestion, foci of hemorrhage and moderate infiltration by lymphocytes and plasma cells. (Figure 3) On these histological findings, a diagnosis of Brunner’s gland hamartoma was made.
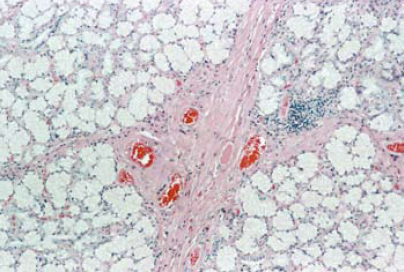
Figure 1a: Lobules of Proliferated Brunner’s Glands separated by Fibromuscular Stroma and Lymphocytic Infiltration with Lymphoid Follicle Formation (H&E x 100).
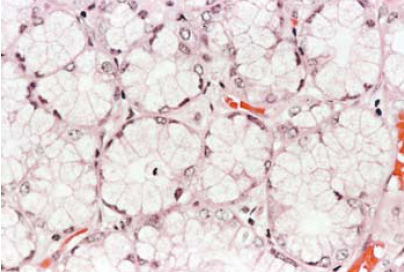
Figure 1b: High power view of Brunner’s glands (H&E x 400).
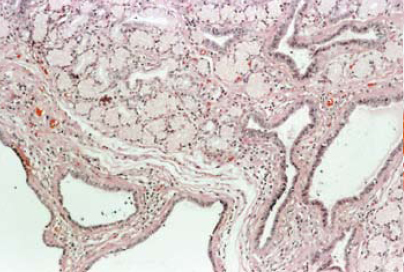
Figure 2: Cystically dilated ducts (H&E x 100).
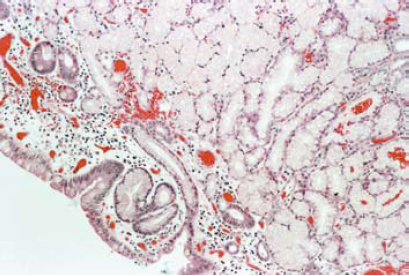
Figure 3: Covering Mucosa showing Congestion and Infiltration by Lymphocytes and Plasma cells with proliferated Brunner’s glands beneath (H&E x 100).
DISCUSSION
Brunner’s glands are branched acinotubular glands located in the submucosa and lined with cuboidal-to-columnar clear cells in the glands, and cuboidal cells in the duct.4 They are predominantly seen in the duodenal bulb and proximal duodenum and progressively decrease in size and number in the distal portion.5 In infants, Brunner’s glands occupy 55% of the total area of the duodenum and by 50 years of age it drops to 35%.6 Functionally, they secrete an alkaline fluid composed of mucin, which exerts a physiologic anti-acid function by coating the duodenal epithelium, protecting it from the acid of the stomach. Furthermore, in response to the presence of acid in the duodenum, they also secrete pepsinogen and urogastrone which inhibit gastric acid secretion.7 Thus, they play a significant role in duodenal resistance to ulcer formation.8
Brunner’s gland hamartoma is a rare benign tumor of the duodenum accounting for about 10.6% of benign duodenal tumors 8 and occurred in about 0.008% of individuals in a single series of 215,000 autopsies.5 Since its first description by Cruveilhier in 1835 and Salvioli in 1876, less than 200 cases have been reported in the literature.2
Most Brunner’s gland hamartomas are located in the duodenal bulb (57%) but may also be found in the second (27%) and third (7%) parts of the duodenum and rarely detected in the pyloric canal (5%), jejunum (2%) and proximal ileum (2%).2 Usually they are pedunculated lesions (89%)1 with size ranging from 1-2 cm. The largest reported was 12 cm in size.8 They are almost always single with only one report of two large lesions in the same patient.2
In the past there has been significant debate regarding the nomenclature of Brunner’s gland proliferations. In 1934, Feyrter categorized the abnormal proliferations into three types: type 1, diffuse nodular hyperplasia, in which multiple sessile projections are found throughout the duodenum; type 2, circumscribed nodular hyperplasia, in which sessile projections are limited to the duodenal bulb; and type 3, glandular adenoma, in which there are polypoid tumor-like projections. It remains a matter of debate whether they represent the spectrum of the same pathological process.7 However, most of the authors prefer to describe lesions less than 1 cm. in diameter as hyperplasia and those more than 1 cm. as adenomas.2,5,8 Hamartoma designates an excessive focal overgrowth of mature normal cells and tissues in an organ, composed of identical cellular elements. Hence, the term Brunner’s gland hamartoma is favored over Brunner’s gland adenoma as histologically they show admixture of normal tissues including Brunner’s glands, ducts, adipose tissue, Paneth cells, mucus glands, pancreatic acini and lymphoid tissue usually without cellular atypia.7,9 They are always benign, though a few cases have been reported in association with epithelial dysplasia, adenocarcinoma and carcinoid tumors. But these reports are exceedingly rare and with unclear clinical relevance.1
The exact pathogenesis of Brunner’s gland hamartoma is still unclear. It is postulated that hyperacidity induces hyperplasia of these glands, but only 45% of patients had increased gastric acid secretion and 20% had gastric hypoacidity.7 Concurrent Helicobacter pylori infection is very common in patients with Brunner’s gland hamartoma, however, their role in the pathogenesis is unclear.8 Another theory suggests that these lesions are a hyperplastic reaction to inflammation as foci of inflammatory cell infiltrate have been found in these. But their mere presence is insufficient evidence for this argument as lymphocytes are normally found in the submucosa throughout the entire length of the gastrointestinal tract.2 Association with peptic ulcer disease, chronic renal failure and chronic pancreatitis has also been described.6
Brunner’s gland hamartoma occurs most commonly in the fifth and sixth decade of life with no gender or race predominance.1 Most of them are asymptomatic and diagnosed incidentally by imaging studies or present with nonspecific vague abdominal pain, discomfort, nausea or bloating. In such cases small polypoid lesions may appear at barium examination or at endoscopy.2 Symptomatic tumors can be divided into two categories viz. hemorrhagic and obstructive. The obstruction occurs when the hyperplasia is diffuse or a single adenoma grows too large, causing epigastric bloating, pain, nausea, vomiting and weight loss.3 The hemorrhagic manifestations due to ulceration or erosion of the tumor are melaena, fatigue, anemia and rarely hematemesis.6 Uncommon presentations include obstructive jaundice, biliary fistula, recurrent pancreatitis or intussusception.7
Diagnosis of Brunner’s gland hamartoma is not always easy at present.5 The differential diagnosis includes gastrointestinal stromal tumor, benign or malignant (previously known as gastrointestinal leiomyoma or leiomyosarcoma), lipoma, endocrine cell tumor (carcinoid tumor), lymphoma, vascular tumor, aberrant pancreatic tissue, adenocarcinoma, adenomyomatous polyp, prolapsed pyloric mucosa, ampullary neoplasm and even a foreign object.4, 8
Investigations by imaging modalities such as barium meal, ultrasonography, CT, and Magnetic Resonance Imaging (MRI) are capable of localizing the tumor. Definite diagnosis can be obtained only by histopathological examination.4 Endoscopy is mandatory as it not only localizes the lesion but also facilitates biopsy.2 However, traditional pinching biopsies usually give a negative result as the tumor is almost always covered entirely with thick, intact mucosa and biopsy forceps are unable to reach the tumoral tissue localized completely in the submucosal layer.3,5 Hence a deeper biopsy is recommended.6
Local excision of the lesion done by either an endoscopic snare cautery technique or excision of the mass via duodenotomy is the main treatment.8 The long term outcome of Brunner’s gland hamartoma resection is favorable, without any reported recurrences of the lesion.7
-
Nes LCF, Ouwehand F, Peters SHA, Boom MJ. A large Brunner’s gland hamartoma causing gastrointestinal bleeding and obstruction. Dig Surg 2007; 24:450-452.
-
Al Khurry LE, Al Khafaji KR, Rakhem SA, Al Rubaie AL, Al Sahili SA. Brunner’s Gland Adenoma at the Gastroduodenal Junction: A Case Report. Arab J Gastroenterol 2006; 7:127-129.
-
Gao YP, Zhu JS, Zheng WJ. Brunner’s Gland Adenoma of Duodenum: A case report and literature review. World J Gastroenterol 2004; 10:2616-2617.
-
Woharndee P, Sornmayura P, Bunyaratvej S. Brunner’s gland adenoma: A report of two cases. J Med Assoc Thai 2005; 88:841-844.
-
Rocco A, Borriello P, De Colibus P, Pica L, Iacono A, Nardone G. Large Brunner’s Gland Adenoma: Case report and literature review. World J Gastroenterol 2006; 12:1966-1968.
-
Chattopadhyay P, Kundu AK, Bhattacharya S, Bandyopadhyay A. Diffuse nodular hyperplasia of Brunner’s gland presenting as upper gastrointestinal haemorrhage. Singapore med J 2008; 49:81-83.
-
Abbas R, Al Kawas FH. Brunner gland hamartoma. Gastroenterology & Hepatology 2008; 4:473-475.
-
Rao VSR, Rayan F, Al Mukhtar A, Stojkovic S, Ahmad SM, Moore PJ. Giant Brunner’s gland adenoma-an unusual cause of gastrointestinal haemorrhage. The Royal College of Surgeons of Edinburgh; 2004.
-
Petersen JM, Felger TS, Goldstein JD. Gastroduodenal intussusception secondary to a giant Brunner’s gland hamartoma. Gastroenterology & Hepatology 2008; 4:471-473.