Non-Hodgkin Lymphoma Mimicking Polymyalgia Rheumatica in a Young Patient
Juma Al-Kaabi, Saeed Ahmed, Azhar Rizvi, Ikram Burney
ABSTRACT
Congenital nasal pyriform aperture stenosis (CNPAS) is a recently defined clinical entity that causes airway obstruction in the neonate as a result of narrowing of the nasal pyriform aperture. The pyriform aperture is the narrowest, most anterior portion of the nasal airway, and a slight decrease in its cross sectional area will significantly increase the nasal airway resistance. This entity should be kept in the differential diagnosis of any neonate or infant with signs and symptoms of upper air way obstruction. The CNPAS presents with symptoms of nasal airway obstruction, which are often characterized by episodic apnea and cyclic cynosis.
Key words: Congenital nasal pyriform aperture stenosis; Neonates.
Submitted: 09 Jan 2008
Reviewed: 27 Feb 2008
Accepted: 21 Mar 2008
From the 1Department of Otolaryngology and Head Neck Surgery, 2Department of Pediatrics, 3Departments of Radiology, Sultan Qaboos University Hospital, Al khod, Oman.
Address correspondence and reprint requests to: Dr Rashid Al Abri, Department of Otolaryngology and Head Neck Surgery, Sultan Qaboos University Hospital, P. O. Box 38, Postal Code 123, Al Khod, Muscat, Sultanate of Oman.
E-mail: ralabri@hotmail.com
INTRODUCTION
Polymyalgia Rheumatica (PMR) is characterized by aching pain in the proximal muscle groups namely the shoulders girdles, pelvic girdles, the neck, and morning stiffness usually for more than one-hour. It is primarily a disease of the elderly and is extremely rare before the age of 50 years. The incidence increases with age, with a peak in people 70-80 years of age. Since PMR can mimic a variety of other rheumatic diseases, the diagnosis can be challenging even in the elderly.1 The main differentials are: Polymyositis (PM), elderly onset seronegative rheumatoid arthritis (RA), late onset spondyloarthropathy, late onset systemic lupus erythematosus (SLE), systemic small vessels vasculitis, and remitting seronegative symmetric synovitis with pitting oedema (RS3PE).2 Further, non musculoskeletal conditions such as, infectious causes (viral illness, endocarditis, tuberculosis), endocrine causes (eg hypothyroidism), malignancy (myeloma, carcinoma and lymphoma) can also mimic PMR. Thus PMR is a diagnosis of exclusion. In younger patients, pure PMR is extremely rare and only a few cases have been reported.3, 4
CASE REPORT
A 26 year old lactating female, was referred to the Rheumatology unit for evaluation of a 2-year history of generalized body aches, involving neck, arms, shoulders, and pelvic girdles muscles. The pain was described as deep aching and intermittent in nature. The history of pain was also associated with weight loss of around 10 kg, low grade fever, general malaise, and minimal morning stiffness. She required non-steroidal anti-inflammatory medications for the relief of her symptoms during this period, but she reported worsening of her symptoms over the two months prior to presentation.
She also had received intramuscular triamcinolone acetate injection, with some relief of symptoms. On examination the patient appeared comfortable, pale, febrile up to 38.5°C. Radial pulses were normal and equal bilaterally, blood pressure was 110/70 mmHg in both arms. There were two small lymph nodes (1cm in diameter) in the left axilla. There was no evidence of muscle weakness or wasting only diffuse tenderness was present over the deltoids and quadriceps muscles. Hip movements were limited on the right side. There was no active synovitis at the peripheral joints. The rest of examination was unremarkable. Laboratory investigations revealed Hemoglobin (Hb) of 8.49 g/dl, microcytic, hypochromic, White blood count (WBC) of 7.9x109/l, absolute neutrophil count 4.41x109/l, absolute lymphocyte count 2.64x109/l, platelet 534x109/l. ESR was 105 mm/FHR and the CRP was 96 (0-8.0 mg/l). Lactate dehydrogenase (LDH) was 583 (91-180u/l), Creatinine kinase (Ck) 21 (26-192 u/l), Uric acid (0.15-0.35 mmol/L), ß-2 microglobulin 2.14 (0.7-1.9 mG/L), anti-nuclear antibody (ANA), anti-nuclear cytoplasmic antibody (ANCA), and rheumatoid factor (RF) were all negative. Hep BsAg, Anti-HCV antibody and HIV were negative.
Although there was no evidence of proximal muscle weakness, EMG was done which was reported as normal. Magnetic Resonance Imaging scan of the thighs did not reveal any signs suggestive of myositis, however, it did reveal hyper-intense signals from the bone marrow suggestive of bone marrow pathology. A bone scan showed widespread high uptake in the peripheral and axial skeleton, suggestive of malignancy. There was also uptake in the right hip joint, which may suggest either synovitis or malignant infiltration. Further imaging with whole body MRI showed extensive marrow infiltration. Bone marrow aspiration revealed two sets of lymphocyte population. However, flow cytometry was no-revealin, and the bone trephine revealed fibrosis with increased reticulin especially in the intra-trabecular spaces, suggestive of infiltration of marrow. The patient under went axillary lymph node biopsy. Histopathology revealed an effacement of the follicular structure of the lymph node with well demarcated lymphoid proliferation, made up of pleomorphic large cells. The malignant cells stained positively with leukocyte common antigen (LCA) and CD 20 antibody, consistent with a diagnosis of diffuse large B cell lymphoma (DLBCL) (Figure 1).
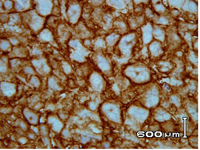
Figure 1: Polymorphic lymphoma cells (CD20 positive cells)
The patient was diagnosed to have stage IVB DLBCL. The patient was started on combination chemotherapy and anti-CD20 antibody, Rituximab (R-CHOP regimen). She tolerated the treatment well and symptoms of pain improved. Re-assessment investigations at the end of 4 cycles revealed a normal bone biopsy, with resolution of fibrosis. The patient received a total of 8 cycles of the combination chemotherapy, at the end of which she was in complete remission. She remains in complete remission for 3 years.
DISCUSSION
In the younger age group, pure PMR should be distinguished from paraneoplastic PMR, or atypical PMR, which could complicate occult malignancy. Features of atypical PMR include age less than 50 years, absence of prolonged morning stiffness, involvement of only one site, ESR <40 or >100 mm/hr, peripheral arthritis, asymmetric involvement at typical sites, and partial or delayed response to steroids.5 Patients presenting with atypical PMR should be investigated for disseminated cancer, connective tissue disease, or different vasculitic disorders such as Takayasu’s arteritis.6, 7
However, the association between malignancy and classical PMR remains unclear. Several prospective and retrospective studies have examined this relationship and increased risk of malignancy was not found in several studies.8-11 However, the low frequency of malignancy seen among these studies could be attributed to prior exclusion of patients with PMR-like symptoms, who are likely to have an underlying malignancy. On the other hand, an increased incidence of malignancy was seen in other studies, which avoided selection of patients.12 Furthermore, several case reports have shown evidence of a relationship, such as, resolution of the symptoms of PMR after treatment of the underlying malignancy.13
Malignancy associated PMR-like manifestations may represent a paraneoplastic phenomenon usually occurring at a site distant from the primary tumor. These manifestations are induced by the tumor with a variety of mechanism such as hormones, peptides, autocrine, paracrine mediators and antibodies. Usually these manifestations can precede the malignancy, appear concomitantly or present later after the diagnosis.5 Alternatively, direct invasion of bones and articular and juxtraarticular structures by the tumor, through metastases, causing release of local inflammatory mediators may be another mechanism. This presentation has been described in several reports and it may be the initial manifestation of widespread malignancy in patients presenting with atypical features. In the case presented here, it is possible that the manifestations were primarily due to the later mechanism.
In NHL, musculoskeletal involvement occurs in about 7-25% of patients some time during the course of the disease.14 Various manifestations have been described such as; polyarthalgia, polyarthritis, bone destruction, and hypertrophic pulmonary osteoarthropathy. PMR-like symptoms can be described but usually in the elderly. To the best of our knowledge this is the first case report of NHL presenting as PMR-like symptoms in a young patient.
The pattern on involvement of the bone marrow with lymphoma is variable.15 Usually there is a generalized involvement; however, the infiltrates may often appear as focal lesions with intervening uninvolved marrow, for example, focal para-trabecular, focal non-paratrabecular, interstitial, diffuse, and sinusoidal of intravascular. Additionally, mixed patterns may also occur. The bone marrow may be involved in up to 25% of the cases of DLBCL at the time of the diagnosis. The degree of marrow replacement correlates with the pattern of infiltration.
With advanced disease, foal lesions enlarge and may coalesce and occupy a greater proportion of the marrow. Alternatively, diffuse involvement is associated with extensive replacement of the normal marrow. In several instances, reticulin is increased and a profound desmoplastic reaction is observed, as was the case in our patient.
CONCLUSION
Our case report demonstrates that PMR-like symptoms (atypical PMR) may manifest in NHL. Although an extensive search for malignancy is not usually needed in patients presenting with the typical or classical PMR, however, search for an underlying malignancy is warranted in patients presenting with atypical PMR. This case demonstrates that one should be aware of such atypical presentation of PMR, thereby enabling earlier diagnosis and treatment.
ACKNOWLEDGMENT
The authors would like to thank Dr. Vasude Rao for the histological images
REFERENCES
-
Gonzalez-Gay MA, Garcia-Porrua C, Savarani C, Olivieri I, Hunder GG. Polymyalgia manifestations in different conditions mimicking polymyalgia rheumatica. Clin Exp Rheumatol. 2000, 18:755-759.
-
Salvarani C, Cantini F, Boiardi L, Hunder G. Polymyalgia rheumatica. Best Practice & Research Clinical Rheumatol 2004; 18:705-722.
-
Nishikawa M, Shouzu A, Imai Y, Inada M. Histocompatibility antigens and polymyalgia rheumatica in a Japanese patient with insulin-dependent diabetes mellitus. Intern Med 1997; 36:935-937.
-
Whittaker PE, Fitzsimons MG. A 24-year-old man with symptoms and signs of Polymyalgia rheumatica. J Fam Pract 1998; 47:68-71.
-
Naschitz JE, Rosner I, Rozenbaum M, Zuckerman E, Yeshurun D. Rheumatic syndromes: clues to occult neoplasia. Seminar Arthritis Rheum, 1999; 29:43-55.
-
Naschitz JE, Slobodin G, Yeshurun D, Rozenbaun M, Rosner I. Atypical Polymyalgia rheumatica as a presentation of metastatic cancer. Arch Intern Med, 1997; 157:2381.
-
Acebes JC, Ibanez J, Tena X, Castaneda S, Rodriguez A, Herrero-Beaumont G. Polymyalgia rheumatica in the young female as a syndrome of presentation of Takayasu’s arteritis. Clin Exp Rheumatol, 1996; 14:223-224.
-
Haga HJ, Eide GE, Burn J, Johansen A, Langmark F. Cancer in association with Polymyalgia rheumatica and temporal arteritis. J Rheumatol, 1993; 20:1335-1339.
-
Chuang TY, Hunder GG, ILstrup DM, Kurland LT. Polymyalgia rheumatica: a 10-year epidemiologic and clinical study. Ann Intern Med, 1982: 97:672-680.
-
Myklebust G, Grant JT. A prospective study of 287 patients with Polymyalgia rheumatica and temporal arteritis: clinical and laboratory manifestations at onset of disease and at the time of diagnosis. Br J Rheumatol, 1996: 35:1161-1168.
-
Bahlas S, Ramos C, Davis P. Clinical outcome of 149 patients with polymyalgia rheumatica and giant cell arteritis. Rheumatol, 1998; 25:99-104.
-
Haugeberg G, Dovland H, Johnsen V. Increased frequency of malignancy found in patients presenting with new onset polymyalgic symptoms suggested to have Polymyalgia rheumatica. Arthritis & Rheumatism (Arthritis Care & Research), 2002; 47:346-348.
-
Niccoli L, Salvarani C, Baroncelli G. Renal cell carcinoma mimicking polymyalgia rheumatica. Scand J Rheumatol, 2002; 31:103-106.
-
McDonald JE, Clarke F, Smith SR, Kesteven P, Walker DJ. Non-Hodgkin’s lymphoma presenting as polyarthritis. Br J Rheumatol, 1994; 33:79-84.
-
Kroft SH, McKenna RW. Bone marrow manifestations of Hodgkin’s and non-Hodgkin’s lymphoma and lymphoma like disorders. In Neoplastic Hematopathology (Ed Knowles DM), 2001. Lippincot Williams and Wilkins. Philadelphia p. 1447-1504.